INTRODUCTION
Central hypersomnolence disorders are excessive daytime sleepiness in the absence of nocturnal sleep pathology or insufficient sleep [
1-
5], including narcolepsy with or without cataplexy and idiopathic hypersomnia (IH) [
6]. Cataplexy is a symptom mainly caused by hypocretin/orexin deficiency and is an essential criterion for classifying narcolepsy types. In this regard, according to the International Classification of Sleep Disorder (3rd edition, 2014), narcolepsy type 1 (NT 1) has cataplexy, abnormally low hypocretin in the cerebrospinal fluid (CSF), and two or more sleep onset rapid eye movement periods (SOREMPs) and the presence of abnormal sleep latency. Narcolepsy type 2 (NT 2) has no cataplexy with normal CSF hypocretin but shows similar multiple sleep latency test (MSLT) results to narcolepsy type 1. Furthermore, IH has no elastic seizures, normal CSF hypocretin, and no SOREMP, but abnormal sleep latency on MSLT [
7].
Since measuring the concentration of hypocretin is an invasive and expensive process with a high risk of infection, the diagnosis of narcolepsy has been mainly based on MSLT [
8]. However, many studies have raised numerous questions about the validity of MSLT. In one study, MSLT was performed again after a mean of 4.2 years for a total of 36 patients diagnosed with type 2 narcolepsy and IH [
6]. Notably, the proportion of diagnosis change was observed to be 53%, specifically 10 out of 15 patients with type 2, and 6 out of 14 patients with IH [
6]. In contrast, Um et al. [
9] reported that different MSLT results occurred only for 2 out of 48 patients diagnosed as NT1 patients in the follow-up MSLT from the initial, most patients satisfying the NT 1 criteria as well. At this moment, it should be noted that these researches had an intrinsic limitation that the number of patients was relatively small.
There have not been many studies on MSLT validity in narcolepsy patients with more than 5 years of follow-up in Korean sleep centers. In this study, we intended to analyze the differences in follow up MSLT parameters of NT1 and NT2, along with their baseline clinical disparities.
We hypothesized that there will be differences in baseline and follow up MSLT between NT1 and NT2 group. Moreover, we hypothesized that the diagnostic changes and clinical course will be disparate between the two groups.
DISCUSSION
This study is one of the longitudinal studies that analyzed the repeated MSLT results in patients with NT1 and NT 2 in the sleep center of the St. Vincent’s Hospital, College of Medicine, Catholic University of Korea. Notably, distinct differences have been demonstrated among the baseline demographic characteristics and the MSLT. Also, in the case of the repeated MSLT parameter, considerable differences were observed for the two groups.
Firstly, our results of the follow-up MSLT findings in NT 2 groups is in same line with the previous studies [
3-
7,
10-
13]. The proportion of maintaining diagnosis in NT2 was only about 50%. Several reasons could be suggested for the sensitivity and specificity of MSLTs. Preferentially, some of the variability resulting from the MSLT may be contributed to methodological problems. Despite the American Academy of Sleep Medicine published clear guidelines for the performance of MSLT, it has been occurred that the specific protocols often differ among sleep labs (i.e., the quantity of the last night’s sleep, timing of the first nap in relation to habitual sleep times and duration, performing 4 vs. 5 naps, optimal nap durations, rigor in ensuring wakefulness among nap opportunities, and methods to score SOREMPs) [
14,
15]. Furthermore, SOREMP is often expressed as false-positive [
13]. In this regard, a previous study reported that subpopulations with excessive sleepiness, including shift workers, young adults with chronic sleep restriction, and patients with sleep apnea, appeared to have a greater prevalence of SOREMP [
13,
16]. In our study, special efforts were made to exclude the cofounders by asking the participants about the confounding factors (i.e., shift workers, having sleep apnea before, etc.).
Secondly, as initially expected, the NT 1 group and NT 2 group showed some differences in the baseline characteristics. NT 1 group demonstrated a larger value of BMI than NT 2 and more sleep paralysis and hypnogogic hallucination, which was consistent with the previous studies [
17]. This difference could be explained by hypocretin/orexin deficiency, which also means NT 1 has nocturnal-sleep wake instability as compared to NT 2 [
17,
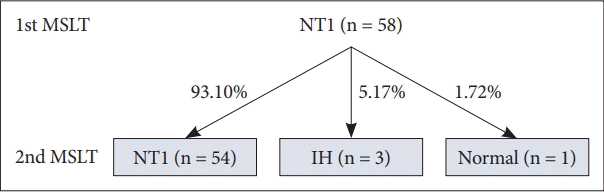
18]. On the other hand, when NT 1 patients repeated MSLT, the proportion of diagnosis retention was greatly high amounting to 93%. It is well known that NT 1 is a well-formed disease, showing a stable course and consistency in nocturnal PSG or MSLT parameters [
7,
19]. Sleep paralysis was also more common in NT 1 group. One study revealed that several symptoms of narcolepsy disorder were frequently present in families associated with patients who suffered from narcolepsy disorder (i.e., simpleplex families with one narcolepsy patient, and multiplex families with two or more narcolepsy patients in families) [
20]. In particular, the incidence and chronicity of hypersomnolence and prevalence of cataplexy-like symptom were observed to be higher in multiplex families than in simple families. Moreover, prevalence of sleep paralysis, incidence and chronicity of hypnagogic hallucination were greater in the multiple family compared to the simple family [
20]. This suggests that the above-mentioned narcolepsy symptoms were more genetically influenced [
20]. Notably, according to our study, BMI, sleep paralysis, and hypnagogic hallucination were significantly higher in NT 1 than in the parallel NT 2 group, with nocturnal sleep disturbance also greater in NT 1, although not statistically significant. From these results, it could be concluded that the five major symptoms of the narcolepsy, which are cataplexy, sleep paralysis, hypnogogic hallucination, hypersomlenonece, and nocturnal sleep disturbance, came out more clearly in NT 1 as compared to the corresponding NT 2 group.
Thirdly, when analyzing the data of follow-up MSLT in narcolepsy patients, we inferred that the change in SOREMP could have a greater effect on the alteration of diagnosis. In all the groups with the change of MSLT, the groups under negative SOREMPs ≥ 2 were observed to be accompanied by negative MSL at follow-up. It should be noted, however, that there is an evident limitation that the number of participants is relatively small. In addition, several studies have placed more emphasis on SOREMP on PSG as a helpful indicator of high specificity to distinguish between narcolepsy and non-narcoleptic central hypersomnia [
21,
22]. In their research, Dietmann et al. [
21] reported that two or more SOREMPs in either two of the MSLT naps or one SOREMP in PSG and at least one SOREMP in MSLT naps had a high sensitivity of 90% and specificity of 95% with a positive predictive value of 91%, thus successfully differentiating narcoleptic from non-narcoleptic patients. According to our data, it was observed that the baseline nocturnal PSG SOREMP in narcolepsy type 2 was 43.8%, and the maintenance rate of narcolepsy type 2 diagnosis was 50%. Indeed, the rest of 50% of the participants at follow-up was led to an alteration in their diagnosis to non-narcoleptic hypersomnia (IH or normal).
Recently, nocturnal PSG has been spotlighted as an alternative option for diagnosing NT1. In one study, following-up of NT 1 and NT 2 patients, respectively, NT 1 patients showed increase in N1 stage in nocturnal PSG compared to NT 2 patients [
17]. Interestingly, some research groups suggested that the sequence of sleep stages on MSLT might be diagnostically helpful, as SOREMPs commonly followed N1 sleep in narcolepsy but not in other disorders [
23,
24]. It was contended that the rapid transition into REM sleep might reflect the REM sleep dysregulation of narcolepsy [
23,
24]. In view of the results above, it is likely that the analysis of MSLT, including sleep stages in nocturnal PSG, may help us diagnose narcolepsy in a more accurate manner.
The limitations intrinsic to this study are as follows: To begin with, our sample size was relatively small, with the sample derived from only one single sleep center, thus making it difficult to generalize the results. Besides, the follow-up MSLT was performed just twice, baseline and follow-up, and follow-up intervals were not the same among patients. Therefore, the results obtained through this research could not reflect the detailed course of parameter changes until follow-up. Moreover, because this study is designed as a cohort study, there is a high probability that patients who have been tested twice in this study had a different course of the disease from in many cases. From this fact, the selection bias could not be excluded. Besides, most of the patients undergoing repeated examinations are taking stimulants or anti-depressants because of the abnormal patterns of REM sleep (cataplexy, hallucinations, nightmares, etc.). Even though we educated patients to stop taking the drugs for more than 2 weeks for the test, it is hard for us to exclude the actual number of patients who continued the drug without reporting to the medical staff during the period. As a result, it is difficult to completely rule out of the drug effect. This study did not statistically compare the two groups of people whose diagnosis changed and those who did not. This is because the number of changes in diagnosis was too small to find a statistically significant, so it seems necessary to do it in a follow-up study. However, all NT 2 patients whose diagnosis has changed failed to meet the SOREMP as much as the diagnostic criteria, so SOREMP will need to be observed more carefully when performing MSLT in the future.
In conclusion, the MSLT measures were observed to be a good diagnostic tool for NT 1 patients, but have some limitations when diagnosing patients suffering from NT 2 because of inconsistency. Therefore, it is crucial to repeat the MSLT regularly to confirm the diagnosis in an accurate manner. Other biomarkers, if available, would be helpful for us to distinguish central hypersomnia. Concurrently, understanding the pathophysiology of type 2 narcolepsy appears to be conducive to accurate diagnosis of the patients. To end, a multi-site prospective study should be helpful to diagnose central hypersomnolence, such as objective measures of sleep history (sleep recordings and actigraphs), circadian phase assessments (dim light melatonin onset), and neuropsychological tests.