A Case of Hypertensive Intracerebral Hemorrhage Accompanying Sleep Apnea
Article information
Abstract
Stroke is very common in patients with sleep disordered breathing, especially in the elderly. We report the case of a 26-year-old man who had been referred to us with a sudden left side motor weakness of the body, headache, chronic fatigue, and witnessed sleep apneas. Intracerebral hemorrhage in the right external capsule and putamen was identified upon brain computed tomography. He had hypertension which had not been diagnosed previously. On polysomnography, apnea-hypopnea index was 73.0/h and arousal index was 74.7/h, indicating severe sleep apnea. Continuous positive airway pressure titration was conducted to determine the optimal pressure to alleviate the respiratory disturbances. Treatment with antihypertensive medication reduced blood pressure (BP) from 197/145 mm Hg to 130/80 mm Hg after 10 days of use. Co-treatment with the medication and auto-adjustable positive airway pressure additionally decreased BP to 110/60 mm Hg and normalized respiratory disturbances. In addition to BP, left hemiparesis, morning headache, daytime sleepiness, and chronic fatigue were all improved. Early treatment of OSA could help facilitate the rehabilitation of or recovery of weakness in such patients.
INTRODUCTION
Stroke is prevalent in patients with sleep-disordered breathing (SDB). About 60% to 70% of all stroke patients are found to have SDB when defined by an apnea-hypopnea index (AHI) of more than 10 per hour.1 In addition, the presence of SDB (AHI ≥ 5) is also associated with a 2 to 4-fold increased risk of developing stroke.2,3 Several studies have shown an independent association between obstructive sleep apnea (OSA) and hypertension (HTN).2,4,5 HTN is regarded as a mechanism which links SDB with stroke. The exact mechanism by which SDB may cause high blood pressure has not been completely elucidated, but it seems to be related to increased sympathetic nerous system activity, sleep fragmentation, intrathoracic negative pressure swings, or impaired arterial broreflex sensitivity.6–8
Intracerebral hemorrhage (ICH) is the most deadly subtype of stroke and is associated with high morbidity and mortality,9,10 as approximately 65% of survivors were found to have died within 1 year.11
A high prevalence of SDB in stroke patients is mainly found among the elderly population (> 65 years of age),12,13 but it is relatively rare in the young age group.
In this study, we present a case of hypertensive ICH in a 26-year-old young man with sleep apnea.
CASE REPORT
A 26-year-old man was referred to the emergency room because of chief complaints of sudden left side motor weakness of the body, which occurred during a class. He also complained of headache, chronic fatigue, and excessive daytime sleepiness. Witnessed sleep apnea was noted by a nurse in the intensive care unit, and the sleep center was consulted for evaluation of sleep apnea. Blood pressure (BP) was reported to be 197/145 mm Hg in the admission examination, but the patient had no history of taking medication for HTN. The physical examination revealed the patient’s weight to be 95 kg, with a height of 172 cm (body mass index = 32.1 kg/m2), and his neck circumference of 40 cm (15.75 inch). He was not a current smoker, but had a history of 3 years. Upon brain computed tomography (CT), he was diagnosed with acute ICH in the right external capsule and putamen by a neurologist (Fig. 1). Brain angiography was further conducted, but no occlusion or stenosis in vessels and no outpouching lesion were found. 2-dimensional echocardiography (M-Mode) revealed mild concentric left ventricular hypertrophy (left ventricular mass index = 124 g/m2) and mild left atrium enlargement (left atrial volume index = 33 mL/m2). On electrocardiogram, normal sinus rhythm with sinus arrhythmia, non-specific T wave abnormality, and prolonged QT were observed. No abnormality in the chest was found by chest X-ray examination. Upon blood chemistry examination, he had higher levels of alanine aminotransferase (88 IU/L), glucose (143 mg/dL), triglyceride (511 mg/dL), and total lipids (1066 mg/dL) than the reference range. Notably, free epinephrine (40.2 ug/day) and norepinephrine (282.5 ug/day) levels were approximately 2-fold and 3.5-fold higher than the upper limit of the reference range, respectively, representing a highly activated sympathetic nervous system in the patient. In nocturnal polysomnography, the AHI was 73.0/h and arousal index was 74.7/h. The most frequently appearing respiratory disturbance was mixed apnea (241 episodes during total sleep period) and then OSA (193 episodes during total sleep period). No central apnea was observed, and the number of hypopnea events was relatively low (5 episodes during sleep period). Continuous positive airway pressure (CPAP) titration was conducted to find the optimal pressure to prevent occurrence of respiratory disturbances. After CPAP titration, AHI was reduced from 73.0/h to 4.3/h and arousal index also decreased from 74.7/h to 15.3/h. Most types of apnea also disappeared, except for central apnea (0 → 2). 9 cm H2O was selected as the optimal pressure.
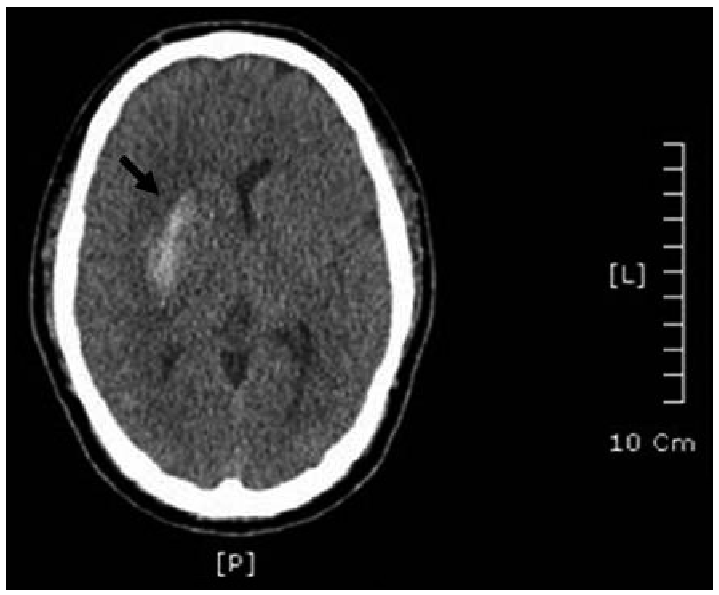
Identification of hemorrhage in the right external capsule and putamen. The hemorrhage is indicated by a black arrow.
Antihypertensive medication was prescribed to reduce BP. After taking the medication for 10 days, the patient’s BP level reduced from 197/145 mm Hg to 130/80 mm Hg (Fig. 2). During 3 days after discharge, auto adjusting-CPAP (APAP) was applied to the patient along with the antihypertensive medication (pressure range: 5–13 cmH2O). APAP is designed to automatically adjust the amount of pressure on a breath by breath basis. It increases compliance by reducing air leaks and noise which can occur at higher pressures. APAP was prescribed instead of CPAP to increase the compliance of the patient, as he had complained of intolerance to the CPAP pressure.

Change of blood pressure (BP) before and after treatment with antihypertensive medication alone and co-treatment with the medication and auto adjusting continous positive airway pressure (CPAP). APAP: auto adjusting-CPAP.
Further reduce of BP to 110/60 mm Hg was observed after combined treatment of APAP and medication. The range of APAP pressure was set from 5 to 13 cmH2O, and the mean pressure recorded during 3 days of treatment was 8.7 cmH2O. At this time, the average AHI was 4.5, indicating almost all respiratory disturbance was successfully abolished by the APAP therapy. The patient was followed-up after 1 month use of APAP. Most of the left hemiparesis was improved. Moreover, body weight and BP were also reduced. As a subjective indicator, morning headache, daytime sleepiness, and chronic fatigue were all improved.
DISCUSSION
Sleep-disordered breathing includes OSA, snoring, upper airway resistance syndrome, and central hypoventilation.14 Among them, obstructive sleep apnea syndrome (OSAS) is the most common type of sleep-disordered breathing, affecting 5–15% of the general population. SDB is highly prevalent in patients with cerebrovascular disease.15 OSA has been observed in up to 77% of patients with acute ischemic stroke, and has been related to increase in the long-term mortality in this population.12,14,16
Sleep-disordered breathing can act as a risk factor of stroke. According to a prospective study from the Wisconsin Sleep Cohort, an AHI index of > 20/h was associated with a 4-fold increased risk of having stroke during a 4-year follow-up study.2 Another prospective study also reported similar results, in which patients with OSAS had a 2-fold higher risk of developing stroke compared with non-OSAS groups over a 3.5 year follow-up, even after adjusting for possible confounders.3
Intracerebral hemorrhage is the most deadly subtype of stroke, and approximately 65% of its surviving victims were found to have died after 1 year.11 OSA, as defined by AHI ≥ 10, was found in 59.4% in 32 non-comatose patients with acute hypertensive ICH.17
The mechanisms underlying cerebrovascular disease in patients with OSA are likely to be multifactorial. Several mechanisms have been considered: hemodynamic disturbances such as HTN, arrhythmias caused by sympathetic activation, endothelial dysfunction, prothrombotic state, and increased inflammatory processes.14
Continuous positive airway pressure therapy is the current treatment used for patients with OSA. The use of CPAP resulted in reductions in systolic BP (−3.20 mm Hg) and diastolic BP (−2.87 mm Hg) according to a meta-analysis study.18 In addition, 1 year of CPAP treatment reduced systolic BP by 1.89 mm Hg and diastolic BP by 2.19 mm Hg in hypertensive patients with sleep apnea,19 possibly by decreasing sympathetic tone and nocturnal hypoxemia, or by preventing pleural pressure fluctuations.8,20
In rare cases, brain damage may contribute to the development of SDB. Cheyne-Stokes breathing can be seen in patients with unilaterial lesions of variable topography without clinically overt heart failure.21,22 Patients with focal lesions in the rostral medulla had reduced ventilatory sensitivity to inhaled carbon dioxide and higher incidence of sleep apnea compared to subjects with no lesions and normal ventilator sensitivity to carbon dioxide. Thus, it could not be determined whether the SDB found in the patient herein was the cause or consequence of the ICH, due to the cross-sectional design of this study. Given that the patient complained of having sleep apnea several years earlier, before the left side hemiparesis became prominent, and that the ICH region was limited to the cerebral region, however, it is reasonable to think that SDB may have affected the development of ICH, although other risk factors should be considered. Re-evaluate of the brain CT was not carried out after APAP, but it is generally known that cerebral hemorrhage is always decreased and eventually absorbed. In addition, hemiparesis usually improves as time goes by. Further study is required to elucidate the degree of contribution of APAP treatment to the improvement in hemiparesis and the size of cerebral hemorrhage in ICH patients accompanied with SDB.
Through this case, we emphasize that the early treatment of OSA could facilitate the rehabilitation of or recovery of weakness in such patients, with some references.
Notes
Conflicts of Interest
The authors have no financial conflicts of interest.