The High-Dose of Exogenous Melatonin Did Not Alter the Sleep-Wake Cycle in Anoxic Brain Injury Patients
Article information
Abstract
Disturbance in circadian rhythms and the sleep-wake cycle is typical for patients in the intensive care unit, which retards rehabilitation. To assess the effect of exogenous melatonin and simultaneous mitigation of intensive care unit environmental factors on sleep duration. We studied five patients with chronic disorder of consciousness caused by anoxic brain injury. In addition, we varied the level of melatonin secretion in blood plasma to assess melatonin’s bioavailability and elimination time. We evaluated the sleep-wake cycle using continuous video-electroencephalogram monitoring with the addition of oculographic and myographic channels for 72 hours. All the patients received melatonin tablets on the second day, wore masks and ear plugs, and had no feeding and nursing manipulations at night on the second and third days. There was no significant difference in sleep time between the first, second, and third days. Future studies of the circadian rhythm should aim at gaining a deeper analysis of the characteristics of the sleep-wake cycle in patients with severe anoxic brain injury together with further research for possible ways to influence the circadian component of sleep.
INTRODUCTION
Anoxic brain injury, which is the diffuse cessation of blood supply to the brain, can be caused by cardiac arrest, massive hemorrhage, asphyxia, and other dysfunctions involving the cardio-respiratory system, often reduces the level of consciousness from a coma, in some cases, to a vegetative or minimally conscious state. In anoxic injury, the vegetative state has an unfavorable prognosis for long-term survival and restoration of consciousness. At the same time, no definitive prognostic markers and effective recovery methods are available [1] .
The concurrent diseases and infectious complications are the usual suspects in the mortality of this group of patients. The factors contributing to these complications in intensivecare units (ICU) are a disturbance in the sleep-wake cycle, decreased total sleep time (TST), fragmentation, and lack of circadian rhythm. The causes for these infringements are various clinical manipulations, such as suctioning of the endotracheal or tracheostomy tubes, hygiene practices, regular change in positions of the patients, and changes in the daylight regime, which in turn decrease the melatonin secretion. In addition, several studies demonstrated decreased melatonin secretion in patients with severe brain injuries, including patients in a vegetative/minimally conscious state [2-5].
The use of exogenous melatonin in ICU patients may increase TST, decrease sleep latency, and improve sleep consolidation [6]. Pavlov et al. [7] showed the connection between the decreased duration of TST in patients with a low level of consciousness and the deterioration of the physical state of the patients; we, therefore, decided to evaluate the effects of exogenous melatonin on patient’s sleep-wake cycle: i.e., the duration of sleep in ICU patients who had undergone severe anoxic brain injury.
MATERIALS AND METHODS
Participants
The study included five anoxic brain injury patients with chronic disorder of consciousness (cDOC) who had undergone treatment and rehabilitation in the ICU. The inclusion criteria were the presence of cDOC caused by severe anoxic brain damage (global ischemia), that is, a vegetative or minimally conscious state. All patients had a minimum of one month of disorder of consciousness after the catastrophic brain injury.
The exclusion criteria were unstable hemodynamics and, consequently, infusion of vasopressors, benzodiazepines, and beta-blockers, and artificial lung ventilation. Two neurologists examined patients independently at various points, made the clinical diagnosis of a cDOC (vegetative or minimally conscious state +/-) and confirmed the diagnosis by a Coma Recovery scale-Revised scale. Medical personnel who had been in contact with the patient were thoroughly interviewed for possible communication with the patient allowed by fluctuations in the level of consciousness.
Written consent was obtained from the medical personnel before the interview and patients as well. In case patient cannot provide the consent the decision was taken by the first degree relative of the patient.
Research Protocol
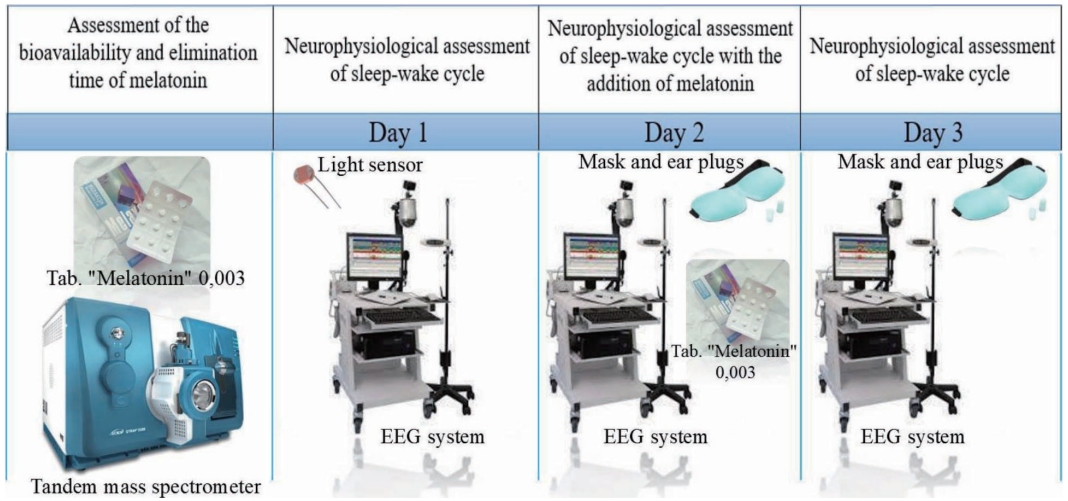
To assess the bioavailability and elimination time of melatonin in the patients, prior to sleep research, we measured the day time blood plasma melatonin level at three points: at the beginning, an hour, and two hours after administering a tablet.
We used video-electroencephalogram (EEG) monitoring with oculographic and myographic channels for three days continuously to find the features of the sleep-wake cycle. We considered longer monitoring to be inappropriate for medical reasons, such as the risk of head pressure sores.
On the first day, video-EEG monitoring was started, and background TST and consolidation of sleep were evaluated. We used a light sensor based on a GL55 series photo resistor (Shenzhen Senba Optical Electronic Co., Ltd. Guangdong, China) to record the illumination in the ICU.
On the second day, patients received a 3-mg melatonin tablet through a nasogastric tube or gastrostomy. We prescribed melatonin to be administered every 3 hours in order to maintain the supraphysiological melatonin concentration in the blood during the day.
Exogenous disturbing factors were limited by the following: 1) wearing sleep masks to limit light pollution at night; 2) inserting earplugs into the ear passages to reduce noise levels; 3) canceling night feeding; 4) limiting nursing manipulations from 22.00 to 06.00.
On Day 2, we also assessed TST and consolidation of sleep via video-EEG monitoring.
Finally, on Day 3, melatonin intake was discontinued, but elimination of exogenous disturbing factors and video-EEG monitoring continued (Fig. 1).
Data Analysis
We measured the melatonin content in blood-serum samples by using tandem mass spectrometry with ultra-efficient liquid chromatography. The method included measuring melatonin in the extract on a tandem mass spectrometer with a triple quadrupole and an AB SCIEX QTRAP 5500 ion trap (AB SCIEX, Concord, ON, Canada) equipped with an atmospheric-pressure chemical ionization source under the control of Analyst software, version 1.6.2 (AB Sciex Pte. Ltd., Woodlands, Singapore). The current of the nebulizer was 2 mA, and the source temperature was 450°C. Melatonin monitoring was carried out in a positive mode, using optimized parameters of ion transitions (multiple reaction monitoring). We used deuterium-labeled melatonin (melotonin-d6) as an internal standard. The measurement results were processed using the MultiQuant 3.0.1 program (AB Sciex LLC, Framingham, MA, USA).
To analyze the EEG data obtained, we formed simplified hypnograms without dividing sleep into phases and stages. The state of “sleep” or “wakefulness” was assigned manually by two neurophysiologists using video data following the international R&K criteria [8] on sleep.
We accumulated, corrected, and systematized the initial information obtained by using the STATISTICA 10 (StatSoft, Inc, Tulsa, OK, USA). We assessed the statistical significance of differences in quantitative traits by using the Wilcoxon W-test. Differences were considered statistically significant at p < 0.05.
RESULTS
Demographic and clinical data of the study participants are presented in Table 1. The median age of patients was 39 [38;49] years.

Demographic data, clinical data, level of melatonin secretion, and sleep time of the study participants
Melatonin level during the daytime without any melatonin tablet supplement was below 1.3 pg/mL in all five patients, which corresponds to the minimum resolution of the method. One hour after patients had ingested 3 mg of melatonin, the average concentration of melatonin in the blood plasma of the sample was 1928.5 pg/mL. After 2 hours, melatonin level was on average 674 pg/mL.
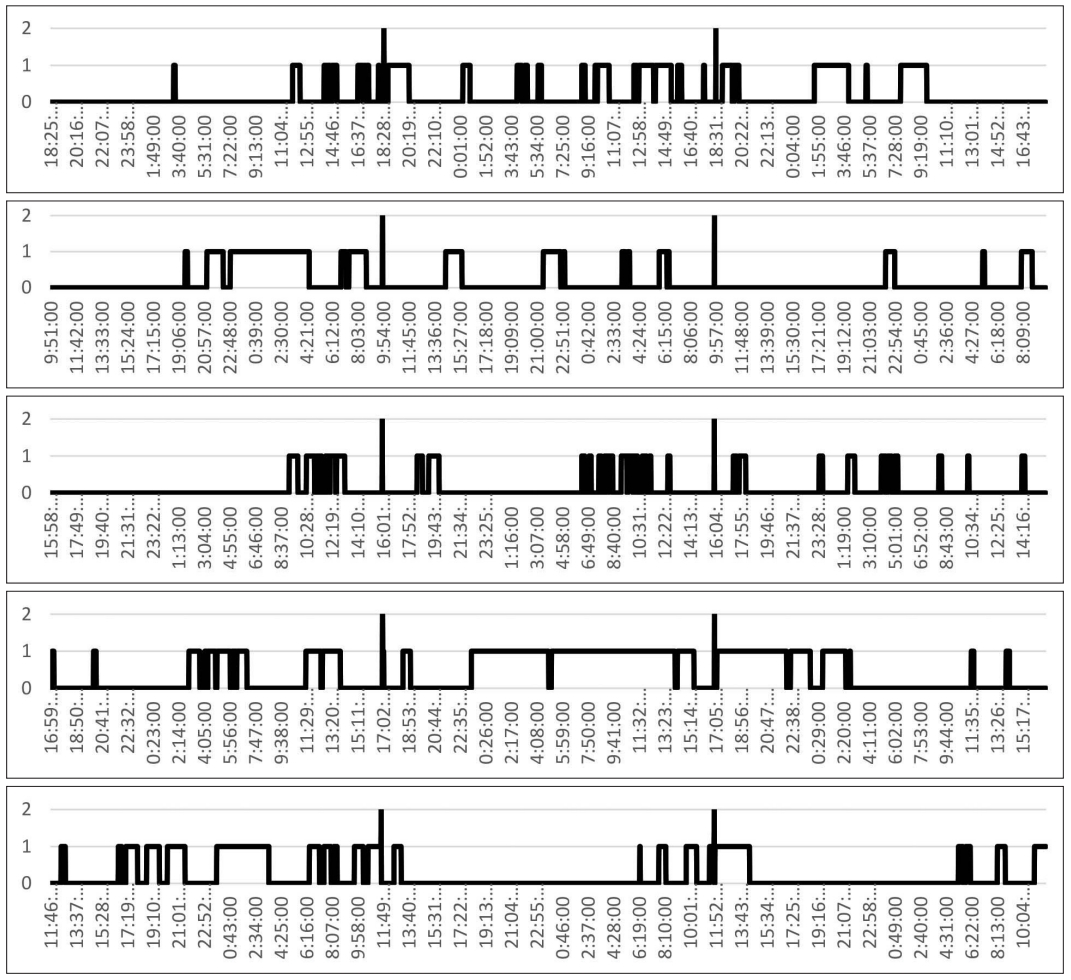
The median value of the TST for 72 hours was 871 [835;1041] minutes (290 minutes per 24 hours), which is significantly lower than that in a healthy population on average. The ratio of the median values of sleep and wakefulness in the first day was 371 [167;522]/1069 [918;1273]; on the second day it was 228 [216; 41]/1212 [1026;1224]; and on the third day, it was 277 [171;333]/1164 [1107;1269]. We found no statistically significant difference in the sleep time between the first, second, and third days (p1-2 = 0.89, p1-3 = 0.68, p2-3 = 0.34).
Consolidation of sleep was weak and did not correspond to night hours (Fig. 2).
Light level in the ICU never got higher than 300 lux and in average constituted 250 lux.
Simplified hypnograms of the study participants are shown in Fig. 2.
DISCUSSION
We used EEG video monitoring to evaluate the sleep-wake cycle in patients with a disorder of consciousness in ICU rather than other assessment methods, such as questionnaires and actigraphy, because patients could not communicate and often had tetraparesis that prevented informative actigraphy signal recording. To assess the homeostatic component of sleep, we used continuous EEG video monitoring for 72 hours. Longer, e.g., weekly, EEG recordings are not possible in immobilized patients with chronic disorders of consciousness, because tissues are injured by prolonged contact with the electrodes.
The decision to give 3 mg of melatonin tablet every three hours between 24 hours and 48 hours, i.e., during the second day of the investigation, was to maintain the supraphysiological concentration of melatonin in blood plasma in order to see if it would affect the sleep. Melatonin per os has good tolerability and scarce evidence of side effects [9,10].
Our study showed that patients with anoxic brain damage and a low level of consciousness had reduced TST during 24 hours than did the healthy population, which multiple reasons can explain. The main reasons are continuous sounds coming from various types of equipment, use of artificial lights, which have lower intensity than that of natural daylight, the constant turning on the lights in the nighttime, regular nursing activities, and nutritional programs with polyphase nutrition. All these reasons play an essential role, which in general can lead to distortion of the circadian component of sleep [11-13]. In addition, diffuse cerebral-cortex injury can also play a role in the sleep-wake cycle because of structural disruption of the integrity of the thalamic-cortico-thalamic circuit’s interactions and a change in the donor-acceptor molecules forming the homeostatic component of sleep [14,15]. Our study did not differentiate the stages of sleep, because anoxic brain damage in patients with a low level of consciousness leads to significant flattening of rhythm, and in some cases to reduction and even elimination of the graphical elements that are signs of a particular stage of sleep, for example, sleep spindles and K-complexes.
Furthermore, there was no significant difference in the amount of sleep on the first, second, and third days even though all participants received 3 mg of melatonin every 3 hours on the second day. Therefore, the melatonin supplement did not have the desired effect on sleep time in our study. We also did not record a difference between the first day, in which there were no restrictions on nursing activities, noise, light, and feeding, and subsequent ones. Although these factors do have a marked effect on the sleep of ICU patients [16], and limiting them may affect the duration and consolidation of sleep in conscious patients [17], the role of external awakening factors in unconscious states is probably questionable.
However, some limitation of our study must be taken into account. First, having only five patients does not allow us to make a general conclusion about patients with severe anoxic brain damage, because are few patients with chronic disorders of consciousness in general and anoxic etiology in particular.
Second, reduced hypnograms make it hard to fully assess the features of the sleep-wake cycle, in particular, to assess the degree of deprivation and return of sleep. In the future, it will be helpful to focus research on the distribution of the third stage of slow-wave sleep (N3 stage non rapid eye movement) to assess sleep quality. It would also be useful to plot changes in melatonin secretion during the day when patients are or are not taking the drug.
Finally, the short observational period of three days resulted from the absence of a pronounced cumulative effect of melatonin. We also did not study the effects of long-term melatonin intake on gene expression.
CONCLUSION
Despite our maintaining a high concentration of melatonin in the blood plasma for two days, we found no significant increase in the TST during the study in anoxic brain-injury patients with a low level of consciousness. Although the hypnotic effect of melatonin is quite small, it can possibly shift circadian rhythms. Therefore, we intend to evaluate the baseline circadian curve and inject exogenous melatonin into patients when the sleep phase starts to maximize consolidation of sleep, which is a major problem for this category of patients.
Future studies also should aim for a deeper understanding of the characteristics of the sleep-wake cycle in patients with severe anoxic brain injury and search for possible ways to influence the circadian component of sleep.
Notes
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Author Contributions
Conceptualization: Mikhail Kanarskii. Data curation: Julia Nekrasova. Formal analysis: Ilya Borisov. Funding acquisition: Marina Petrova. Investigation: Pranil Pradhan, Angelina Nikitkina. Methodology: Pranil Pradhan. Project administration: Marina Petrova. Resources: Marina Petrova. Software: Ilya Borisov. Supervision: Marina Petrova, Olga Korepinа. Validation: Ekaterina Kondratyeva. Writing—original draft: Julia Nekrasova, Mikhail Kanarskii. Writing—review & editing: Julia Nekrasova.
Conflicts of Interest
The authors have no potential conflicts of interest to disclose.
Funding Statement
None
Acknowledgements
We would also like to extend our special thanks to all the members of the intensive care units for facilitating our work.