Treatments for Adult Obstructive Sleep Apnea
Article information
Abstract
Obstructive sleep apnea (OSA) is a chronic sleep disorder that requires a long-term multidisciplinary approach. OSA is characterized by repetitive upper airway collapses during sleep, resulting in various symptoms and outcomes. Therefore, accurate diagnosis and appropriate treatment are needed. In general, positive airway pressure (PAP), oral appliance (OA), and surgery are considered as the primary therapeutic methods, and weight loss and positional therapy are additional or adjunctive treatment options. PAP is recommended for the standard treatment of moderate-to-severe OSA [apnea-hypopnea index (AHI) ≥ 15] and is the optional treatment for mild OSA (5 ≤ AHI < 15). OA is indicated in mild-to-moderate OSA (5 ≤ AHI < 30) that favors OA over PAP. Surgical treatment is usually recommended for patients who manifest anatomical defects that can be alleviated surgically or show a high degree of surgical success rate. Surgery is also indicated in cases refractory to or contraindicated for medical treatments such as PAP and OA. In addition, surgical therapy may be performed as adjunct treatments along with other therapies. Diverse therapeutic options including the indications, mechanisms of action, clinical effects, and limitations should be discussed with the patient during the counseling sessions. Furthermore, the patients should actively participate in the treatment of their own disorder. In summary, an individualized therapeutic method should be considered based on a comprehensive analysis of the patient’s physical findings, such as upper airway anatomy, and obesity, results of polysomnography, such as AHI, respiratory disturbance index, and minimum oxygen saturation, in addition to therapeutic preferences and expectations.
INTRODUCTION
Obstructive sleep apnea (OSA) in adults should be evaluated as a chronic disease that needs long-term and comprehensive care [1]. OSA is characterized by repeated events of complete or partial upper airway collapse during sleep [2].
The symptoms and signs of OSA are numerous, including habitual snoring, observed apnea, morning headache, excessive daytime sleepiness, fatigue, memory loss, and decreased libido, etc. [3]. OSA should be diagnosed accurately based on history, physical examination, upper airway evaluation, for example, using imaging tests and drug-induced sleep endoscopy, and polysomnography [4]. Untreated OSA increases the risk of serious complications, such as hypertension, myocardial infarction, angina, arrhythmia, stroke, type 2 diabetes, and decreased sexual function [5-7]. Therefore, patients diagnosed with OSA require prompt and effective treatment.
The therapeutic modalities for OSA include positive airway pressure (PAP), oral appliance (OA), surgery, weight loss, and positional therapy. PAP, OA, and surgery are considered primary treatments, and weight loss and positional therapy represent adjunctive treatments [1,8]. There is no single treatment that addresses the challenges of all OSA patients at once. Since the indications, effects, advantages, and disadvantages associated with each therapeutic method differ, appropriate customized management is essential for each patient [9-11]. In order to determine the therapeutic option appropriate for each patient, the patient’s physical findings, polysomnographic outcomes, and opinion should be considered comprehensively [1,8]. It is not necessary to insist on the first or single treatment method alone. If the first therapeutic option is ineffective or fails, another management option can be applied. In addition, two or more treatments may be combined for successful management.
This article will review the features of various therapeutic methods including indications, mechanisms of action, clinical effects, and limitations and will provide useful insight into the therapeutic principles for OSA.
THERAPEUTIC METHODS
Positive Airway Pressure
PAP is primarily recommended for the treatment of moderate-to-severe OSA [apnea-hypopnea index (AHI) ≥ 15] [1,12,13]. PAP is also optionally indicated for the management of mild OSA (5 ≤ AHI < 15) [1,12,13]. Therefore, PAP represents a substantial treatment of choice for all types of OSA regardless of the severity of AHI. PAP supplies pneumatic splint to maintain the patency of the upper airway in patients with OSA and as pressure increases, the upper airway tends to enlarge [14,15]. PAP therapy has various positive effects clinically [16]. Based on high-level evidence-based studies, PAP treatment effectively decreases AHI and improves subjective and objective measures of excessive daytime sleepiness [16,17]. In addition, PAP therapy improved quality of life and decreased high blood pressure in low-level evidencebased studies [16,18,19]. According to the recent the American Academy of Sleep Medicine clinical practice guideline for PAP, PAP is recommended for the management of adult OSA with excessive daytime sleepiness [20]. PAP is also suggested for the treatment of adult OSA with decreased sleep-related quality of life or comorbid hypertension [20]. Moreover, PAP is effective in reducing the risk of various adverse events including motor vehicle accidents [21]. However, PAP is inconvenient and associated with side effects such as unintentional mask removal, pressure intolerance, air or oral leakage, skin injury, nasal obstruction, rhinorrhea, nasal bleeding, and aerophagia [1,16,22,23]. Consequently, these adverse effects can lead to decreased PAP compliance.
Oral Appliance
OA is recommended for the management of mild-to-moderate OSA (5 ≤ AHI < 30) in patients who prefer OA over PAP or for those who are intolerant or refractory to other treatments including PAP, positional therapy, and weight loss [1,24-27]. In addition, OA is indicated for the reduction of simple snoring that cannot be managed by other therapies such as change of sleep position or weight control [1,27]. OA increases the patency of upper airway during sleep in OSA patients via the following mechanisms: 1) expansion of the upper airway and/or; 2) diminished upper airway collapse or increased upper airway muscle tone [1,28]. According to numerous studies (mostly low-level evidencebased studies), OA therapy significantly improves AHI in OSA that is usually not severe and alleviates various clinical symptoms and signs including excessive daytime sleepiness [28-31]. In addition, OA treatment has beneficial but weak effects on daytime systolic and diastolic blood pressure and decreases daytime heart rate compared with inactive or placebo methods [32,33]. OA has several limitations such as dental-related side effects and discomfort. Side effects associated with OA usage include excessive salivation, dry mouth, tooth and myofascial pain, gum irritation, temporomandibular joint disorder, dental misalignment, and occlusal change [1,27,28,34]. These limitations can reduce the compliance with OA.
Surgery
There are three indications for the surgical treatment of OSA. The primary indication for surgery in patients with OSA is an obvious obstructive abnormality of the upper airway that can be improved surgically or a high probability of successful surgical outcomes, such as surgery for severe obstructing upper airway mass, and tonsillectomy for large tonsils [1,8]. Second or alternative indication involves cases who are contraindicated for non-surgical therapies including PAP or OA [1,8]. Third, surgery is indicated as an adjunctive treatment to increase tolerance of other OSA therapies, for example, nasal surgery to improve PAP or OA compliance due to nasal obstruction [1]. Most surgeries improve OSA via mechanisms that surgically modify or reconstruct the upper airway anatomy to enlarge the space and/or increase muscle tension [1,8,35]. OSA-related surgeries consist of the following: nasal surgeries, such as septoplasty, turbinate surgery, nasal polypectomy or tumor removal, and endoscopic sinus surgery; nasopharyngeal surgeries, such as adenoidectomy and nasopharyngeal tumor removal; oral and oropharyngeal surgeries, such as uvulopalatopharyngoplasty (UPPP) and modifications, tonsillectomy, and palatal implants; hypopharyngeal surgeries, such as tongue base reduction, lingual tonsillectomy, genioglossus advancement, hyoid suspension, and epiglottoplasty; laryngeal surgeries, such as laryngeal polypectomy or tumor removal; and multi-level or other surgeries, such as oropharyngeal and hypopharyngeal surgery, maxillomandibular advancement, tracheotomy, and bariatric surgery [1,8,35,36]. Nasal surgery plays several important roles in OSA patients. According to a recent meta-analysis, isolated nasal surgery appears to decrease AHI, but only with minimal significance [37]. In addition, it is associated with reduction of subjective and objective signs and symptoms of snoring, improvement of excessive daytime sleepiness, a decrease of therapeutic pressure in PAP users, and increased PAP compliance [38-40]. UPPP is generally one of the most common surgical procedures in patients with OSA. UPPP is not associated with a high rate of surgical success when performed randomly, whereas it increases the surgical success rate when performed selectively based on anatomical staging [41,42]. Multi-level surgery is associated with a higher surgical success rate than the single-level surgery when two or more obstruction sites are suspected [43]. Transoral robotic surgery for base of tongue reduction is mostly performed via palatal surgery, and the results are comparable to those of multi-level surgery [44]. Maxillomandibular advancement is a surgical procedure that extends the retropalatal and retrolingual airway by advancing the maxilla and mandible simultaneously, reducing AHI comparable to PAP in most patients [45,46]. Hypoglossal nerve stimulation is a recently developed surgical management procedure using an implanted neurostimulation system that increases upper airway muscle tone to prevent the collapse of the pharynx and decreases AHI moderately [47,48]. Surgery may yield diverse clinical outcomes including symptoms and signs, quality of life, cardiovascular risk, motor vehicle accidents, and mortality [49-51]. However, surgical limitations include uncertainty in predicting surgical success, surgical risks, and complications, such as foreign body sensation, dryness, velopharyngeal incompetence, speech alterations, and swallowing abnormalities [1,8,52,53].
Weight Loss
Weight loss is recommended for the treatment of all overweight or obese OSA patients [1,8,54]. Weight loss should be combined with a primary treatment such as PAP, OA, and surgery [1,54]. Obesity increases the volume of upper airway soft tissue, narrowing the upper airway, and the upper airway fat deposition increases the collapsibility, resulting in OSA with worsening outcomes [55]. Successful weight reduction via dietary management may alleviate AHI in patients with obese OSA [1,54]. According to a longitudinal study, a weight gain of 5%, 10%, or 20% may lead to an approximate increase of 15%, 32%, or 70% in AHI, respectively [56]. However, a weight loss of 5%, 10%, and 20% is expected to result in approximately 14%, 26%, and 48% reduction in AHI, respectively [56]. Weight loss has several limitations including the reduced success rate of dietary control, and the low cure rate by dietary restriction alone [1,54]. Bariatric surgery is indicated for the management of obese patients who carry a body mass index (BMI) ≥ 40 kg/m2 or a BMI ≥ 35 kg/m2 with significant obesity-related co-morbidities, such as hypertension, type II diabetes, lipid abnormalities, non-alcoholic fatty liver disease, and osteoarthritis, and have been resistant to treatment via dietary weight loss [57]. Bariatric surgery should also be considered as a minimally invasive therapy for use as an adjunct therapy to active and first-line interventions such as PAP [1,54].
Positional Therapy
Positional therapy is recommended for the management of patients with OSA exhibiting AHI that is more than double in the supine position compared with the non-supine sleep position [1,54,58]. Positional therapy can be used as an adjunct to primary treatment in OSA patients and performed using a positioning device, such as alarm, ball, pillow, vest, or vibrator [1,54,59,60]. Positional therapy improves OSA by avoiding the supine position: the upper airway soft tissue including soft palate and tongue are placed inferiorly under the effect of gravity, leading to a decrease in the upper airway size or patency and resulting in increased respiratory disturbances such as apnea or hypopnea during sleep [58,59]. According to the related meta-analysis of 21 papers, positional therapy significantly improves AHI by 54.1% and the lowest oxygen saturation by 3.3%, whereas it does not significantly affect arousal index or sleep efficiency [60]. Positional therapy is associated with several limitations. It is basically difficult to maintain a patient in one position (non-supine position) during sleep without side effects [1,54]. A few established or standardized positional therapeutic devices have been used with objective monitoring [1,54]. Further, positional therapy does not normalize AHI in all OSA patients [1,54].
THERAPEUTIC PRINCIPLES
There are three important points to consider when treating adult OSA [1,8]. First, OSA is considered a chronic disease requiring prolonged and multidisciplinary approach to management such as hypertension, diabetes, and allergic disease. Second, the patients should be actively involved in determining and contributing to their therapeutic options. Third, the most appropriate OSA therapy should be based on the patient’s physical findings, such as upper airway anatomy and obesity, in addition to the results of polysomnography such as AHI, respiratory disturbance index (RDI), and minimum oxygen saturation, along with the patient’s therapy preferences and expectations. The therapeutic principles for OSA are summarized in Fig. 1.
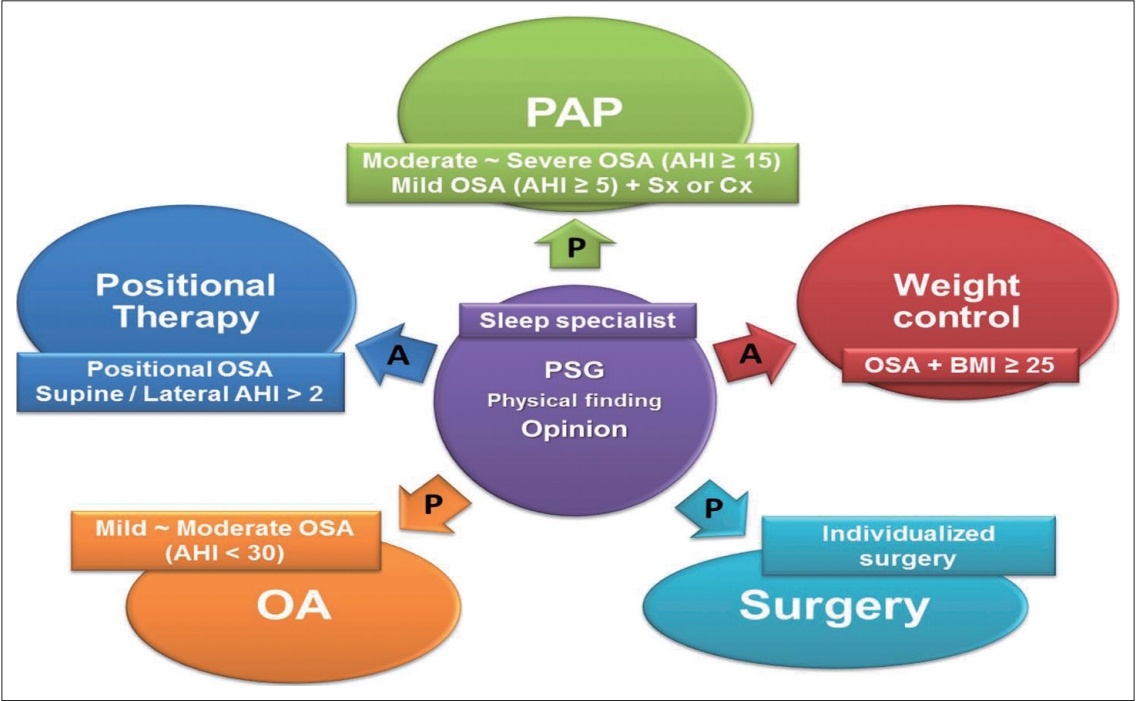
Therapeutic principles underlying OSA. PAP: positive airway pressure, OA: oral appliance, OSA: obstructive sleep apnea, AHI: apnea-hypopnea index, Sx: symptoms, Cx: complications, BMI: body mass index, P: primary treatment, A: adjunctive treatment, PSG: polysomnography.
Patient’s Physical Findings (Anatomy)
Before deciding the treatment approach, the patient’s physical findings should be evaluated [1]. Physical findings include general appearance such as obesity, and increased neck circumference; nasal cavity, such as deviated nasal septum, hypertrophied turbinate, and nasal polyp or tumor; features of nasopharynx, such as adenoid vegetation and nasopharyngeal tumor; characteristics of oral cavity and oropharynx, such as high arched palate, large soft palate, elongated uvula, hypertrophied palatine tonsil, and macroglossia; hypopharynx, including hypertrophied lingual tonsil, omega-shaped epiglottis, hypopharyngeal tumor, micrognathia, and retrognathia; and larynx, including paralyzed vocal cords and laryngeal polyps or tumors [61,62].
Patient’s Polysomnography
Important polysomnographic parameters that are useful in determining the treatment method include sleep data, including the total recording time, total sleep time, sleep efficiency, sleep latency, wake after sleep onset, and sleep stages; arousal parameters, such as arousal index, respiratory arousal index, and spontaneous arousal index; respiratory parameters, such as AHI, RDI, oxygen desaturation index ≥ 3% or 4%, mean oxygen saturation, minimum oxygen saturation, and snoring; cardiac parameters such as average heart rate during sleep, highest heart rate during sleep, bradycardia, and tachycardia; and movement-related parameters, such as periodic limb movements of sleep (PLMS) index and PLMS arousal index [63].
Patient’s Opinion
The outcomes of physical examination and polysomnography are explained to the patient. The sleep specialist also explains an individualized natural course of disorder, risk or aggravating factors, risk factor modification, and possible consequences [1,8]. In addition, various OSA therapies including the indications, mechanisms of action, clinical effects, and limitations associated with each method should be counseled [1,8]. Further, the patients should actively participate in the management of their disorder, expressing their opinions including therapeutic expectations and preferences [1,8].
CONCLUSION
Personalized therapeutic options should be considered systematically according to the patients’ physical findings, including upper airway structure and obesity; polysomnographic outcomes, such as AHI, RDI, and minimum oxygen saturation; and therapeutic preferences and expectations.
Acknowledgments
This study was supported by the Soonchunhyang University Research Fund.
Notes
Conflicts of Interest
The author has no financial conflicts of interest.
