AbstractBackground and ObjectiveThis study aimed to characterize patterns of hypnotic drug prescription based on age and gender using data from the Health Insurance Review and Assessment Service-National Patients Sample (HIRA-NPS) of South Korea.
MethodsUsing HIRA-NPS data, the ŌĆ£number of tablets of equivalent hypnotic drugs (TEQ)ŌĆØ was defined herein as the number of tablets of triazolam, zolpidem immediate-release form, or zolpidem extended-release form.
ResultsThe proportion of hypnotic medication prescriptions, including triazolam and zolpidem, was 1.47% in the sample population. The proportion was significantly higher among women than men (1.63% vs. 1.29%; p < 0.01). In addition, the proportion of subjects who were prescribed hypnotic drugs increased with age. The mean TEQ per year was significantly higher among women in the 30ŌĆō39 year old group (p = 0.02), but lower in women of the 70ŌĆō79 year old group compared to men (p = 0.04). Of the subjects prescribed the drugs, 14.53% were prescribed at least 0.5 TEQ per day, while 5.0% were given at least 1.0 TEQ per day 0.6% at least 2.0 TEQ per day. Such cases were most common among middle-aged patients (30ŌĆō59 years old).
INTRODUCTIONInsomnia is a commonly reported clinical symptom. The annual prevalence of insomnia symptoms has been reported to range from 35ŌĆō50% of the population,1 including cases of insomnia disorders occurring at 10ŌĆō22%,2,3 although the exact frequency depends on the diagnostic criteria applied. The prevalence of insomnia symptoms in Korea was reported to be 17ŌĆō23%,4,5 including Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition diagnosis in 5% of the patients.4 Insomnia is predictive of psychiatric disorders, including depression, anxiety disorders, and alcohol or drug abuse, and is also associated with physical function and quality of life. The prevalence of insomnia is greater in the elderly population.4,5 Insomnia shows a female predisposition which is consistent and progressive with age, becoming more prevalent in the elderly.6 Additionally, it has been reported that chronic benzodiazepine or hypnotic drug users are most often elderly.7,8 The pharmacokinetics and clearance of hypnotic drugs may be altered in elderly individuals compared to younger individuals. It is well known that benzodiazepine, which has a long half-life, may be associated with an increased incidence of adverse events, such as confusion or falling down, among the elderly.9ŌĆō11
Several hypnotic drugs have been approved by the Korean Food and Drug Administration (KFDA). Physicians can also prescribe many hypnotic medications for insomnia without KFDA indication, including antipsychotics, benzodiazepine, and sedative antidepressants such as amitriptyline and trazodone antihistamine. Many people who suffer from sleep problems do not discuss them or their use of hypnotic drugs with their doctors. Currently, over-the-counter medications and alcohol are widely used as sleep aids. The potential for overtreatment with hypnotic drugs without proper sleep hygiene education raises health concerns in the population.12 This study aimed to explore the patterns of hypnotic drug prescription according to age and gender using data from the Health Insurance Review and Assessment Service-National Patients Sample (HIRA-NPS).
METHODSPatientsData from the HIRA-NPS-2011 (serial number: HIRA-NPS-2011-0101) were analyzed to explore patterns of hypnotic medication prescription in South Korea. This study was approved by the Institutional Review Board of Asan Medical Center. The HIRA-NPS-2011 is a stratified random sampling for gender and age from the entire population enrolled in the Korean National Health Security System from January 2011 to December 2011. The total sample size was 1375832 people, which is 3% of the entire recorded patients.
Calculating the Number of Tablets of Equivalent Hypnotic drugs (TEQ)Herein, the ŌĆ£number of tablets of equivalent hypnotic drugs (TEQ)ŌĆØ was defined as the quantity of triazolam, zolpidem immediate-release form, or zolpidem extended-release form tablets. Numerous studies have shown that 0.25 mg triazolam is comparable to 10 mg zolpidem in the immediate-release form,13ŌĆō15 and that 10 mg of zolpidem in the immediate-release form is comparable to 12.5 mg of zolpidem in the extended-release form.16,17 The TEQ was calculated by summing the number of tablets of 0.25 mg triazolam, 10 mg zolpidem immediate-release form, or 12.5 mg zolpidem extended-release form prescribed per day. For example, if a subject was prescribed 0.125 mg of triazolam, 5 mg of zolpidem immediate-release form, or 6.25 mg of zolpidem extended-release form, the TEQ would be 0.5. If a subject was prescribed 0.25 mg of triazolam and 5 mg of zolpidem immediate-release form at the same time, the TEQ would be 1.5. The TEQ/year was calculated by summing the total number of hypnotic tablets prescribed in one year. If a subject was prescribed 0.125 mg triazolam for 30 days and 10 mg zolpidem immediate-release form for 30 days in a year, the TEQ/year would be 45.
Statistical AnalysesThe Stata program version 13.1 (STATA Corp, College Station, TX, USA) was used to select subjects who were prescribed any hypnotic drug. Patient medical records were merged (using a unique encrypted identification number) and the daily and annual TEQ was calculated, The StudentŌĆÖs t-test for TEQ and the chi-square test for the proportions of subjects who were prescribed hypnotic drugs were then employed for sex and age group comparisons. All statistical tests were two-tailed, and results were considered to be significant with p values < 0.05.
RESULTSSubjects Prescribed Hypnotic Drugs at Above 3 TEQ Per DayFrom HIRA-NPS, 27 subjects (18 women and 9 men) were identified who were prescribed hypnotic drugs at above 3 TEQ per day during 2011. These 27 cases included 2 women in the 20ŌĆō29 year old age group, 2 men and 7 women in the 30ŌĆō39 year old age group, 2 men and 1 woman in the 40ŌĆō49 year old age group, 1 man and 4 women in the 50ŌĆō59 year old age group, 3 men and 1 woman in the 60ŌĆō69 year old age group, and 1 man and 3 women in the 70ŌĆō79 year old age group. Among these subjects, three were prescribed an unusually high amount of hypnotic drugs (3351, 5156, and 5305 TEQ) during 2011. These cases are further described below.
Case 1A 26-year-old woman was prescribed 3351 tablets of zolpidem over 2011. On several occasions, she was prescribed 100 tablets of zolpidem over 5ŌĆō8 day intervals.
Case 2A 56-year-old man was prescribed 5156 tablets of triazolam in 2011. During this period, he was prescribed 100 tablets of triazolam for 21 and 28 days, respectively, and one tablet of triazolam per day for several months.
Case 3A 57-year-old woman was prescribed 5305 tablets of hypnotic drugs (596 zolpidem and 4706 triazolam tablets) in 2011. This included prescriptions of 100 tablets of triazolam for 7, 14, and 21 days, respectively, 1 tablet of triazolam daily for several months, and 1 tablet of zolpidem daily for several months.
Patterns of Hypnotic Drug PrescriptionAmong the 1375832 subjects in the HIRA-NPS dataset for 2011, subjects under 10 and over 100 years old were excluded from statistical analyses. The 27 subjects who were prescribed more than 3 TEQ per day were also not included in the final analyses. Among the total HIRA-NPS cohort, hypnotic medications, including triazolam and zolpidem, were prescribed to 18,306 subjects (1.47%)(Table 1). The proportion of females who were prescribed hypnotic medication was significantly higher than males (1.63% vs. 1.29%; p < 0.01). The proportion of females who were prescribed hypnotic medication was significantly higher than men in the 30ŌĆō79 year old groups, but lower in the 80ŌĆō99 year old group. In the 80ŌĆō89 year old group, 5.5% of subjects were prescribed hypnotic drugs (Fig. 1). The rate was also high among the elderly population (70ŌĆō99 years old). Triazolam was prescribed to 5152 HIRA-NPS subjects (0.41%) and zolpidem was prescribed to 14874 (1.19%) subjects. Among the men, triazolam was prescribed to 2063 (0.34%) and zolpidem to 6453 (1.08%) subjects. Among the women, triazolam was prescribed to 3089 (0.48%) and zolpidem to 8421 (1.30%) subjects.
The mean TEQ per year was significantly higher among women in the 30ŌĆō39 year old group (p = 0.02), but significantly lower in the 70ŌĆō79 year old group (p = 0.04) compared to men (Table 1). The proportion of subjects who were prescribed at least 0.5 TEQ per day was 14.5% (Fig. 2), and high rates were observed among the 40ŌĆō49 (17.2%), 50ŌĆō59 (16.4%), and 30ŌĆō39 (15.9%) year old groups (Table 2). The proportion of subjects who were prescribed at least 1.0 TEQ per day was 5.0%, and 0.6% received at least 2.0 TEQ per day. These instances were highest among the middle-aged (30ŌĆō59 year old) groups.
DISCUSSIONIn our current analyses, we observed the hypnotic drug prescription rate of 1.47% in South Korea, with the highest prevalence among the elderly. However, the total number of medications taken annually was found to be highest in 30ŌĆō39 year old females and 70ŌĆō79 year old males. Among patients who took any of the hypnotic medications, 5.0% took at least one hypnotic drug tablet every day annually.
Numerous studies have shown that the prevalence of insomnia is higher in the elderly,4,5 among whom it can often be a prodromal symptom for an underlying physical and/or mental illness.18 In addition, the elderly are more likely to take benzodiazepine hypnotics.19 Psychological problems, comorbid medical conditions, decreased physical activity, multiple usage of medications, and pharmacokinetics are contributors to the higher prevalence of insomnia and use of hypnotic drugs in the elderly.20 In the current study, a high proportion of elderly subjects (70ŌĆō99 years old groups) (Fig. 1) were observed to take hypnotic medication, which is in agreement with previous studies. However, the TEQ per year and the proportion of subjects who were prescribed at least one hypnotic drug tablet per day were higher among the 30ŌĆō59 year old groups (Table 1, 2). We conclude from this that the elderly tend to take hypnotic drugs more frequently, but at a lower dosage.
In this study, the proportion of females who were prescribed hypnotic medication was significantly higher than males among the 30ŌĆō79 year old groups. Numerous studies have reported the prevalence of insomnia to be higher in women,6,16 and that older women tend to use sedatives or hypnotics more frequently.21 Women are more vulnerable to stressful events or psychological stresses. Insomnia is a common complaint in various psychiatric disorders, especially depression and anxiety.22 In addition, greater bodily vigilance and awareness is found among women, and a socially sanctioned culture exists that encourages women to more readily express emotional distress and somatic symptoms.23 Nevertheless, the proportion of the current study subjects who were prescribed hypnotic drugs was highest among men in the 80ŌĆō99 year old group. This result could be a consequence of the low sample size of male subjects in this age group, or the exclusion of other hypnotic agents such as trazodone or various benzodiazepines from this study, which might have skewed the results. However, this finding indicated that much older men tend to take hypnotic drugs more often.
Three subjects in the HIRA-NPS cohort for 2011 were found to have been prescribed ŌĆ£unusuallyŌĆØ high amounts of hypnotic drugs for an unknown reason. However, we speculate that these individuals did not use the hypnotic drugs appropriately or just for sleep issues alone. Numerous studies have reported evidence of zolpidem abuse and dependence.24ŌĆō26 In addition, history or abuse and underlying psychiatric diseases can increase the risk of zolpidem abuse.27 Hypnotics impair cognition, memory, and performance, and also increase the risk of car accidents.28 In addition, the sleep-promoting effects of hypnotics appear to be diminished with chronic use because of a build-up of tolerance.29 In chronic insomnia patients for whom pharmacotherapy is unsuccessful, comorbid mental and physical factors indicate a sleep disturbance complexity that is unlikely to respond fully to medication.30 We conclude from these findings that careful attention to the hypnotic drug use patterns is needed to prevent drug abuse and dependence.
In the present study, only the prescription rates for triazolam and zolpidem were estimated, and other hypnotic medications such as trazodone or various benzodiazepines were not included. Benzodiazepines, including clonazepam, are commonly used for sleep problems, while trazodone is widely prescribed for maintenance of sleep. It was not determined in this study whether trazodone or benzodiazepine was prescribed in the HIRA-NPS cohort for sleep problems or other symptoms, such as pain, anxiety, or movement disorders. This may have skewed our findings on the prescription rates of hypnotic agents. In addition, only the 2011 data from the HIRA-NPS was used, thus the long term use of hypnotic medications was not explored. Despite the limitations of this study, however, our findings demonstrated that the proportion of hypnotic drug prescriptions increased with age and was more common in females. Furthermore, the level of hypnotics used annually was greatest among males and in the middle-aged (40ŌĆō59 years old) group of patients.
REFERENCES1. Walsh JK, Coulouvrat C, Hajak G, Lakoma MD, Petukhova M, Roth T, et al. Nighttime insomnia symptoms and perceived health in the America Insomnia Survey (AIS). Sleep 2011;34:997-1011.
2. Roth T, Coulouvrat C, Hajak G, Lakoma MD, Sampson NA, Shahly V, et al. Prevalence and perceived health associated with insomnia based on DSM-IV-TR; International Statistical Classification of Diseases and Related Health Problems, Tenth Revision; and Research Diagnostic Criteria/International Classification of Sleep Disorders, Second Edition criteria: results from the America Insomnia Survey. Biol Psychiatry 2011;69:592-600.
3. Morin CM, LeBlanc M, Daley M, Gregoire JP, M├®rette C. Epidemiology of insomnia: prevalence, self-help treatments, consultations, and determinants of help-seeking behaviors. Sleep Med 2006;7:123-30.
4. Ohayon MM, Hong SC. Prevalence of insomnia and associated factors in South Korea. J Psychosom Res 2002;53:593-600.
5. Cho YW, Shin WC, Yun CH, Hong SB, Kim J, Earley CJ. Epidemiology of insomnia in Korean adults: prevalence and associated factors. J Clin Neurol 2009;5:20-3.
7. Rumble R, Morgan K. Hypnotics, sleep, and mortality in elderly people. J Am Geriatr Soc 1992;40:787-91.
8. Bertisch SM, Herzig SJ, Winkelman JW, Buettner C. National use of prescription medications for insomnia: NHANES 1999ŌĆō2010. Sleep 2014;37:343-9.
9. Moloney ME, Konrad TR, Zimmer CR. The medicalization of sleeplessness: a public health concern. Am J Public Health 2011;101:1429-33.
10. Greenblatt DJ, Harmatz JS, von Moltke LL, Wright CE, Durol AL, Harrel-Joseph LM, et al. Comparative kinetics and response to the benzodiazepine agonists triazolam and zolpidem: evaluation of sex-dependent differences. J Pharmacol Exp Ther 2000;293:435-43.
11. Rosenberg J, Ahlstr├Ėm F. Randomized, double blind trial of zolpidem 10 mg versus triazolam 0.25 mg for treatment of insomnia in general practice. Scand J Prim Health Care 1994;12:88-92.
12. Wesensten NJ, Balkin TJ, Belenky GL. Effects of daytime administration of zolpidem versus triazolam on memory. Eur J Clin Pharmacol 1995;48:115-22.
13. Barkin RL. Zolpidem extended-release: a single insomnia treatment option for sleep induction and sleep maintenance symptoms. Am J Ther 2007;14:299-305.
14. Greenblatt DJ, Legangneux E, Harmatz JS, Weinling E, Freeman J, Rice K, et al. Dynamics and kinetics of a modified-release formulation of zolpidem: comparison with immediate-release standard zolpidem and placebo. J Clin Pharmacol 2006;46:1469-80.
15. Eaton WW, Badawi M, Melton B. Prodromes and precursors: epidemiologic data for primary prevention of disorders with slow onset. Am J Psychiatry 1995;152:967-72.
16. Stewart R, Besset A, Bebbington P, Brugha T, Lindesay J, Jenkins R, et al. Insomnia comorbidity and impact and hypnotic use by age group in a national survey population aged 16 to 74 years. Sleep 2006;29:1391-7.
18. Greenblatt DJ, Harmatz JS, Shader RI. Clinical pharmacokinetics of anxiolytics and hypnotics in the elderly. Therapeutic considerations (Part I). Clin Pharmacokinet 1991;21:165-77.
19. Spore DL, Mor V, Larrat P, Hawes C, Hiris J. Inappropriate drug prescriptions for elderly residents of board and care facilities. Am J Public Health 1997;87:404-9.
20. Glass J, Lanct├┤t KL, Herrmann N, Sproule BA, Busto UE. Sedative hypnotics in older people with insomnia: meta-analysis of risks and benefits. BMJ 2005;331:1169.
21. Wysowski DK, Baum C. Outpatient use of prescription sedative-hypnotic drugs in the United States, 1970 through 1989. Arch Intern Med 1991;151:1779-83.
22. Piccinelli M, Wilkinson G. Gender differences in depression. Critical review. Br J Psychiatry 2000;177:486-92.
23. Barsky AJ, Peekna HM, Borus JF. Somatic symptom reporting in women and men. J Gen Intern Med 2001;16:266-75.
24. Victorri-Vigneau C, Dailly E, Veyrac G, Jolliet P. Evidence of zolpidem abuse and dependence: results of the French Centre for Evaluation and Information on Pharmacodependence (CEIP) network survey. Br J Clin Pharmacol 2007;64:198-209.
25. Heydari M, Isfeedvajani MS. Zolpidem dependence, abuse and withdrawal: A case report. J Res Med Sci 2013;18:1006-7.
26. Quaglio G, Faccini M, Vigneau CV, Casari R, Mathewson S, Licata M, et al. Megadose bromazepam and zolpidem dependence: two case reports of treatment with flumazenil and valproate. Subst Abus 2012;33:195-8.
27. Hajak G, M├╝ller WE, Wittchen HU, Pittrow D, Kirch W. Abuse and dependence potential for the non-benzodiazepine hypnotics zolpidem and zopiclone: a review of case reports and epidemiological data. Addiction 2003;98:1371-8.
28. Lai MM, Lin CC, Lin CC, Liu CS, Li TC, Kao CH. Long-term use of zolpidem increases the risk of major injury: a population-based cohort study. Mayo Clin Proc 2014;89:589-94.
Fig.┬Ā2.Equivalent hypnotic drug tablets prescribed per year. Each open circle represents one patient. The 27 subjects who were prescribed 3 or more tablets of equivalence hypnotic drugs per day were not included in this figure. TEQ: tablets of equivalent hypnotic drugs. 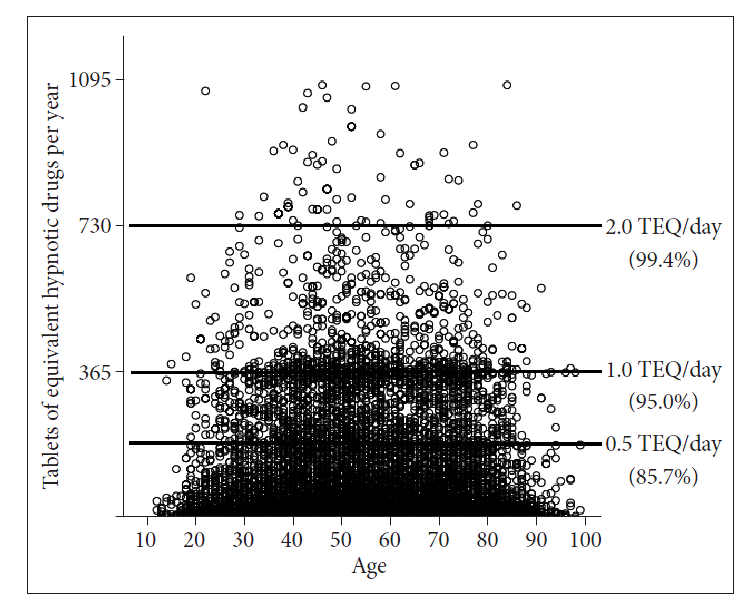
Table┬Ā1.Proportion of subjects prescribed a hypnotic drug (triazolam or zolpidem) in 2011 and the calculated TEQ
Table┬Ā2.Number of tablets of equivalent hypnotic drugs (TEQ) prescribed by age group |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||