AbstractDue to glossal and craniofacial skeletal abnormalities, children with Down syndrome (DS) are prone to obstructive sleep apnea (OSA), thus demanding an increased necessity for an earlier diagnosis and more strict control for OSA. Adenotonsillectomy (T&A op) is an effective surgical treatment method to reduce OSA in children. Nevertheless, it should be noted that T&A op in DS children might be insufficient to completely resolve OSA and that extra care is needed when performing a T&A op for airway obstruction in DS during the immediate postoperative period. The objective of the present study was to report a case of a 2-year-old male DS patient with severe OSA (apnea-hypopnea index [AHI] of 61.7/hr) which was consequently reduced to moderate OSA (AHI of 7.6/hr), followed by reduction to mild OSA (AHI of 4.8/hr) with an one-year of rapid maxillary expansion (RME) device application. With a 2-year application of the RME device, the hard palate width in the 1st and 2nd molar region was increased to 5.86 mm and 5.26 mm, respectively. This is the first case report describing that a severe OSA is reduced to moderate OSA with T&A op and further reduced to mild OSA with an RME device application.
INTRODUCTIONDown syndrome (DS) is a chromosome disorder resulting from trisomy in the chromosome 21. Due to soft tissue and skeletal alterations, DS children are prone to upper airway obstructive disorders, including obstructive sleep apnea (OSA) which has been reported to be seen in half of DS preschool children [1]. Untreated OSA in DS children may lead to the development hypoxia following pulmonary hypertension (PH) [2]. Therefore, OSA in DS children should be assessed more thoroughly and managed more strictly.
For pre-school children with snoring and sleep apnea, adenotonsillectomy (T&A op) is an effective surgery frequently performed worldwide for many decades. Nevertheless, DS children bear the risk for life-threatening upper airway complication with T&A op in intraoperative and early postoperative periods [3]. Hence, surgeons should be aware of these special conditions when performing T&A op in DS patients and pay an additional attention to intraoperative and postoperative periods.
Here, the authors report a case of OSA in 2-year-old male DS child showing serial reductions of apnea-hypopnea index (AHI) scores after T&A op followed by rapid maxillary expansion (RME) device application.
CASE REPORTA 2-year-old male patient visited the outpatient office of an ear, nose, and throat (ENT) physician with severe snoring and sleep apnea. The patient had a previous history of confirmed DS with genetic study. The patient also presented with a frequently recurring bilateral otitis media with effusion, which resulted in the insertion of a ventilating tube. On physical examination, symmetric palatine tonsillar hypertrophy graded as grade II bilaterally with a moderate degree of adenoid vegetation assessed with the lateral paranasal sinus X-ray image was noticed. On level I polysomnography (PSG) scored by a board-certified trained sleep medicine specialist, a total AHI of 62.4/hr was assessed. As recommended by the international classification of sleep disorders 3rd edition published by the American Academy of Sleep Medicine [4], we applied the AHI in the pediatric population as sum of obstructive and mixed AHI when assessing the AHI in the pediatric population. Therefore, an initial AHI of 61.7/hr was confirmed for this child (Table 1).
Continuous positive airway pressure (CPAP) titration was not possible because the child could not tolerate this procedure. Therefore, automatically controlled positive air pressure device was tried with levels ranging from 4 to 10 cm H2O. However, the patient’s guardian resigned to use CPAP device in a few weeks complaining very low compliance in this severely uncooperative child. Hence, the authors recommended T&A op as the second therapeutic option for resolving OSA in this child. Upon scheduling an elective T&A op in this child, we were fully aware of the possibility of severe life-threatening upper airway complications in the immediate postoperative period. Therefore, we booked a pediatric intensive care unit (PICU) for immediate postoperative care with a bedside preparation for an urgent intubation or tracheotomy. The patient was ordered to apply the CPAP device in the immediate postoperative period to secure the upper oropharyngeal airway which might be more narrowed due to postoperative soft tissue edema in the oro-velopharyngeal area.
The patient underwent an elective T&A op under general anesthesia without intraoperative complication. Postoperatively, the patient was transferred to the PICU overnight and then transferred to the general ward on postoperative day (POD) 1. The CPAP device was applied in the immediate preoperative period until POD 5. The patient was discharged on POD 6. The patient did not develop any complications (i.e., bleeding, airway obstruction, etc.) in the postoperative period.
In the 6-month postoperative period, the child’s snoring had improved. However, his mother complained an occasional snoring in the supine position upon sleeping. The POD 6-month PSG revealed a reduction in the AHI, showing an AHI of 7.6/hr (Table 1). The severe OSA in the preoperative period was dramatically improved to a moderate degree OSA. However, the authors felt the necessity of an additional intervention for a more strictly reduced OSA owing the fact that the child was confirmed with DS.
Since the patient’s parents still reported that the child could not bear the discomfort of the CPAP device, the authors have consulted a board-certified orthodontist (Y. J. Kim) for the application of an RME device in this child in the POD 6-month period. A tooth-support type RME device with metal band was customed and applied for this patient with an intention to expand the hard palate horizontal width as well as the maxilla as elaborated in a previously published article [5]. The RME device was intended to widen the hard palate bone for the first three weeks, followed by an adjustment period which the osteogenesis would take place to fill the gap in between the hard palate bone up to 9 months. Afterwards, the patient was asked to apply the fixed RME device for maintenance purpose. At the 2-year visit, a horizontal expansion of the palate bone width was noticed (Fig. 1A and B). The increased palate bone widths in the first and second molar were 5.86 mm and 5.26 mm, respectively (Fig. 1C).
At one year after initiation of the RME device application, the patient’s PSG revealed an AHI score of 4.8/hr, showing a mild degree of OSA (Table 1). Additionally, the patient’s guardian reported a diminished snoring even in supine position during the child’s sleep. The patient was scheduled for a yearly visit to the ENT specialist and orthodontist’s office.
The requirement of informed consent was waived, as approved by the Institutional Review Board of the Asan Medical Center.
DISCUSSIONIn this case report, the authors present a gradual reduction of OSA with T&A op followed by RME device application in a DS confirmed 2-year-old child. It has been reported that about 66% to 95% of DS children present with OSA. The incidence of OSA in the infant era has been reported to be up to 71% in DS children [6]. DS children present with variable abnormalities. They especially show a short neck, an enlarged and protruded tongue, a narrower and more highly-arched hard palate, and hypotonia, all of which contributes to the development and increased severity of OSA [6].
In DS children, cardiac anomalies and PH are frequently associated [2]. On the other hand, prolonged hypoxemia itself works as an independent risk factor for PH development. Numerous reports have discussed negative effects of OSA in the pathophysiology of PH [2]. Therefore, OSA in the DS population should be more strictly regulated and physicians should understand the necessity of prompt recognition of OSA along with suggestion of a more comprehensive discipline plan for the management of OSA.
T&A op is one of the most frequently selected treatment option to reduce OSA and snoring in the pediatric population. It has been recognized to show effectiveness in reducing OSA and OSA-associated complications in children over many decades [7]. Nevertheless, when performing T&A op in DS children for the treatment of OSA, the ENT surgeon must bear the following three considerations.
First, the surgeon must be aware of possible upper airway obstruction due to immediate postoperative oropharyngeal swelling which may aggravate the airway narrowing already narrowed by anatomical anomalies (i.e., macroglossia, short neck, increased volume in the cervical soft tissue) in DS patients. Additionally, DS children receiving T&A op show a perioperative respiratory adverse event of about 20% [3]. To prevent such postoperative catastrophe, all measures to secure the upper airway patency in the immediate postoperative ear should be settled before heading into the surgery while managing the patient in the PICU setting as recommended by other authors [3].
Second, during the T&A op, the patient’s neck should be extended to guarantee maximal visualization of the palatine tonsil and adenoids. However, the atlanto-axial joint instability frequently accompanied in DS patients brings a risk for atlantoaxial joint dislocation during the surgery [6].
Third, the ENT surgeon should keep in mind that the OSA in DS children might not be fully managed only with T&A op itself and that some degree of OSA or snoring may persist even after complete recovery from the T&A op, which was also shown in our case. Therefore, it is important to assess and reassess the severity of OSA by repetitive PSG in the postoperative period. In case of remaining OSA, additional multidisciplinary treatment strategy should be encouraged with the consultation to each specialist [1], as demonstrated in the current case.
In children with persisting OSA following T&A op, the application of CPAP could serve as a treatment option [8]. However, poor compliance of the CPAP device especially in children brings the necessity for another treatment option. Mandibular advancement device (MAD) is also a treatment option for OSA [9]. However, it is not a permanent treatment option. In addition, it requires frequent device changes along with very limited indication.
On the other hand, RME can widen the horizontal width of the narrowed hard palate [10], which is a characteristic finding of OSA patients, thereby expanding the oropharyngeal airway in addition to widening of nasal cavity. With an enlarged oropharyngeal airway, the retro-positioned tongue is relocated anteriorly, enabling a reduction in the airway resistance in the oropharynx and reducing the OSA [10]. RME is relatively simple, safe, and manageable in the office setting. It can be recommended as a therapeutic option especially in patients with syndromic children having congenital anomalies in the craniofacial region [10].
As shown in the current case, management of OSA in preschooler DS children might be challenging as these children are more likely to be not cooperative with CPAP device. With a multidisciplinary team approach, the current case highlights the importance of a need for a permanent and sustainable treatment protocol for a stricter reduction of OSA in DS children.
In conclusion, the anatomical difference in DS children brings a need for a more comprehensive disciplines for the management of comorbid OSA. The authors would like to emphasize that the ENT surgeon should be fully aware of possible complications during the intraoperative or postoperative period in DS children upon T&A op. As far as we know, the current case report is the first study in the literature to describe a severe OSA down staged to moderate OSA with T&A op, which is consequently reduced to mild OSA with an additional intervention with an RME device.
NOTESAvailability of Data and Material
All data generated or analyzed during the study are included in this published article.
Author Contributions
Conceptualization: Marn Joon Park, Yoo-Sam Chung. Data curation: all authors. Formal analysis: Marn Joon Park, Yoon-Ji Kim. Investigation: Marn Joon Park, Yoo-Sam Chung. Methodology: all authors. Project administration: Yoo-Sam Chung. Resources: Yoo-Sam Chung, Yoon-Ji Kim. Software: Yoon-Ji Kim. Supervision: Yoo-Sam Chung. Validation: Yoo-Sam Chung, Yoon-Ji Kim. Visualization: Marn Joon Park, Yoon-Ji Kim. Writing—original draft: Marn Joon Park. Writing—review & editing: Marn Joon Park, Yoo-Sam Chung.
REFERENCES1. Maris M, Verhulst S, Wojciechowski M, Van de Heyning P, Boudewyns A. Prevalence of obstructive sleep apnea in children with Down syndrome. Sleep 2016;39:699-704.
2. Loughlin GM, Wynne JW, Victorica BE. Sleep apnea as a possible cause of pulmonary hypertension in Down syndrome. J Pediatr 1981;98:435-7.
3. Xiao L, Barrowman N, Momoli F, Murto K, Bromwich M, Katz SL. Risk factors for respiratory adverse events after adenoidectomy and tonsillectomy in children with down syndrome: a retrospective cohort study. Eur J Pediatr 2022;181:2399-408.
4. Sateia MJ. International classification of sleep disorders-third edition: highlights and modifications. Chest 2014;146:1387-94.
5. Haas AJ. Palatal expansion: just the beginning of dentofacial orthopedics. Am J Orthod 1970;57:219-55.
6. Goffinski A, Stanley MA, Shepherd N, Duvall N, Jenkinson SB, Davis C, et al. Obstructive sleep apnea in young infants with Down syndrome evaluated in a Down syndrome specialty clinic. Am J Med Genet A 2015;167A:324-30.
7. Dempsey JA, Veasey SC, Morgan BJ, O’Donnell CP. Pathophysiology of sleep apnea. Physiol Rev 2010;90:47-112.
8. Chawla J, Forwood C, Heussler H. CPAP to treat obstructive sleep apnoea (OSA) in children with Down syndrome (DS). Eur Respir J 2016;48:PA3066.
Fig. 1.A 3D cast scan model of maxillary teeth and hard palate before and at two years following rapid maxillary expansion (RME) treatment. A: A 3D cast scan model of maxillary teeth and hard palate bony structure prior to the application of RME device. B: A 3D cast scan model following two years of RME device application. C: A superimposed image of initial and final 3D cast scan model. Note the expanded width of the hard palate is 5.86 mm in the 1st molar region and 5.26 mm in the 2nd molar region. 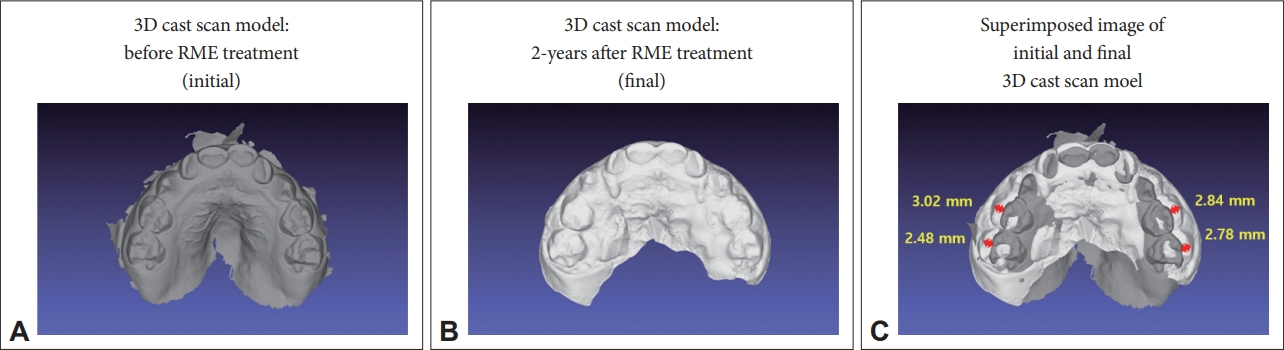
Table 1.Changes in clinical and PSG parameters following T&A op and RME treatment
* Total AHI–central AHI was described to represent the obstructive sleep apnea/hypopnea in a child as recommended by the international classification of sleep disorders 3rd edition published by the American Academy of Sleep Medicine [4]. |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||