AbstractBackground and ObjectiveHeart rate vatiability (HRV) parameters are altered during sleep in patients with obstructive sleep apnea (OSA). Nondipping of nocturnal heart rate during sleep is characteristic, but not all patients with OSA show this. The aim of this study was to identify differences in OSA patients with or without nocturnal heart rate dipping.
MethodsWe retrospectively reviewed patients (n = 40) who were confirmed to have OSA [4% Oxygen desaturation index (ODI4) > 5/hr) by a simplified sleep study and underwent a 24 hours Holter electrocardiogram. The OSA patients were compared with control subjects (n = 72), who had been found to have no significant medical diseases.
ResultsMost HRV parameters were lowered in OSA patients [standard deviation of all normal R-R intervals (SDNN): 99.23 vs. 149.06 ms: p < 0.0001]. The OSA patients were divided to two subgroups according to their 24 hours HR tachogram patterns. A HR nondipper subgroup (n = 18) showed a marked decrease in SDNN when compared with a HR dipper subgroup (n = 22)(SDNN 71.72 vs. 121.72 ms: p < 0.0001). SDNN was not associated with a severity index of OSA (ODI4)(r = ŌłÆ0.15, p = 0.3619).
ConclusionsIn the OSA patients, most HRV parameters (SDNN, standard deviation of the average normal R-R intervals for each 5-min period (SDANN), percentage of adjacent cycles that are > 50 ms apart (pNN50%), root mean square successive differences in milliseconds were lower than in the control subjects. The OSA patients can be divided to two subgroups according to a 24 hours HR tachogram patterns (HR dipper and nondipper). In HR nondipper OSA subgroup, HRV parameters (SDNN, SDANN), Mean SaO2 and basal heart rate during sleep were markedly lowered, but were not associated with an OSA severity (ODI4).
INTRODUCTIONThe disappearance of nocturnal physiological heart rate dipping has been observed in patients with obstructive sleep apnea (OSA), and this phenomenon might suggest the existence of underlying OSA.1 Nondipping of nocturnal (that means during sleep) heart rate may be due to disturbed sleep or fragmentation of sleep, which is a characteristic of OSA, but not all OSA patients show this tendency. We therefore attempted to identify clinical differences in OSA patients with or without nocturnal heart rate dipping.
METHODSWe retrospectively reviewed patients (n = 40, male 35, female 5) who were confirmed to have OSA [4% Oxygen desaturation index (ODI4) > 5/hr] by Win-Visi monitoring system (Stowood Scientific Instrument, Oxford, UK) and had undergone 24 hours monitoring with a Holter electrocardiogram (Marquette Series 8000 Holter Scanner, Milwaukee, USA). The patients with OSA and a comparison group of control subjects (n = 72, male 40, female 32) were recruited from medical records of a tertiary general hospital. The control subjects had been found to have no significant medical diseases. The 24 hours heart rate tachogram pattern was taken from a Holter electrocardiogram (Marquette Heart Rate Variability Software Version MARS 4.1B). A heart rate dipper pattern was defined as the dipping of nocturnal heart rate more than 10% from baseline level before sleep. Heart rate vatiability (HRV) parameters were derived from the Holter monitoring data. Comparison of data between patients with OSA, control subjects and OSA subgroups were done with T test using Med-Calc 11.4 statistical program (Mariakerke, Belgium).
RESULTSAs expected, when we compared the parameters of OSA patients with those of the control subjects, OSA patients showed lower HRV values such as mean value of all normal-to-normal interbeat intervals (Mean NN), standard deviation of all normal R-R intervals (SDNN), standard deviation of the average normal R-R intervals for each 5-min period (SDANN), percentage of adjacent cycles that are > 50 ms apart (pNN50%), root mean square successive differences in milliseconds (rMSSD) (Table 1). The basal heart rate during sleep (SHR) taken from the 24 hours heart rate tachogram also showed a marked differ ence with the OSA group displaying significantly higher SHR values than the controls (Table 1). When the OSA group was divided into two separate subgroups based on their 24 hours heart rate tachogram pattern (HR nondipper vs dipper) (Figs. 1 and 2),2 the HR nondipper subgroup (n = 18) was found to be significantly different from the HR dipper subgroup (n = 22) in SDNN, SDANN, ratio of low to high frequency (LHR), Mean SaO2, and SHR (Table 2 and 3)(Figs. 3, 4 and 5). The two sub groups did not differ in terms of ODI4, pulse rate rise (PRR) > 6/min, Epworth sleepiness scale, Body mass index, or neck circumference (Table 2 and 3). The results of a correlation analysis are shown in Table 4. In the undifferentiated OSA group, SD-NN was found to be significantly related to both SHR and Mean SaO2, but it was not associated with Mean SaO2 in the HR dipper subgroup. SHR was related to most HRV parameters (Mean NN, SDNN, SDANN, pNN50%, and rMSSD) in patients with OSA, but in the HR dipper subgroup, it was unrelated to rMSSD, pNN50, ODI4, PRR and Mean SaO2. In contrast, SHR was significantly associated with ODI4, PRR, Mean SaO2, MeanNN, SDNN, SDANN, pNN50%, and rMSSD in the HR nondipper subgroup.
DISCUSSIONAlmost all energy on earth is derived from the sun. On our planet, biological systems have evolved to develop sophisticated and efficient processes of energy utilization.3ŌĆō6 Efficiency of energy utilization is reflected on the interaction of heart and lung, or ŌĆścardiopulmonary couplingŌĆÖ. We can detect and measure the efficiency of cardiopulmonary coupling in many ways. HRV analysis is one well known method of evaluating cardiopulmonary coupling. HRV is known to be decreased in OSA,7 but increased HRV in OSA has been reported.8,9 The HRV pattern during sleep has revealed pathological, as well as physiological changes. The pathological changes induced by sleep disordered breathing may therefore increase HRV parameters, especially in severe OSA.8,9 A daytime decrease in HRV may reflect sympathetic overactivity as a carry-over effect, so the 24 hours HRV pattern is more important than night time HRV. Contrary to a previous report,7 the severity index of OSA (ODI4) was not associated with SDNN, which is the best index of HRV(r = ŌłÆ0.15, p = 0.3619). Certain other parameters of HRV were not associated with ODI4: these were SDANN, rMSSD, and pNN-50%. ODI4 was related only to MeanNN and LHR. The two sub groups were different in the association with ODI4 and Mean-NN:, the relationship held in HR nondipper subgroup (r = ŌłÆ0.57, p = 0.0142), but not in HR dipper subgroup (r = ŌłÆ0.11, p = 0.6150). SDNN had significant relationship with Mean SaO2 (r = 0.56, p = 0.0002) in the undifferentiated OSA group, wher eas this relationship was found in the HR nondipper subgroup (r = 0.69, p = 0.0017). In the HR dipper subgroup, there was no association with Mean SaO2 (r = 0.08, p = 0.7096). This finding may suggest the possibility of different prognoses, but a long term follow study will be necessary to prove this is the case. It may be thought that in the HR dipper subgroup, better SDNN (121.73 vs. 71.72 ms), Mean SaO2 (93.58 vs. 90.98%) and lower SHR (64.41 vs. 81.56/min) already suggested a more preserved homeostatic condition. SDNN showed the highest association with SHR (r = ŌłÆ0.81, p < 0.0001) in both the subgroups of HR nondipper and dipper. SDNN is a very sensitive predictor of morbidity and mortality,10 and resting heart rate is strongly related to the risk of sudden cardiac death.11 It is therefore not surprising that SDNN was closely related to SHR in our study (r = ŌłÆ0.81, p < 0.0001). This may be deduced from the finding that HR and SDNN have shown reciprocal changes simulating mirror images.12 The division of OSA patients into HR dipper and nondipper subgroups, which is very simple and easy by 24 hours heart rate tachogram, showed marked differences in SDNN, SDANN, SHR and MeanSaO2, but no differences in ODI4 and PRR. The nocturnal HR dipping pattern had no association with ODI4 or, PRR. This finding may suggest the possibility that HR dipping during sleep is a kind of endophenotype.13
In the long term, many of the HRV parameters (SDNN, SD-ANN, SHR and Mean SaO2) are expected to be related to cardiovascular mortality and morbidity, but further studies will be necessary to confirm this. Our study suggest that the 24 hours heart rate tachogram pattern may give clinically significant informations about subgroup differentiation (HR dipper vs. non-dipper) and resting basal heart rate (i.e., sleep heart rate: SHR), which showed markedly different HRV pattern.
Limitations. This was not prospective study, and sampling biases are therefore a particular risk. Moreover the diagnosis of OSA was confirmed by a simplified sleep study (Win-Visi monitoring system), and the results of this may be different from those using traditional polysomnography.
ConclusionIn OSA patients, most HRV parameters (SDNN, SDANN, pNN-50%, and rMSSD) showed lower levels than in the control sub jects. When OSA patients were divided to two subgroups according to 24 hours HR tachogram patterns (HR dipper vs. nondipper), HRV parameters (SDNN, SDANN, Mean SaO2, and SHR) in the HR nondipper subgroups were markedly lowered, but were not associated with the OSA severity index of ODI4.
REFERENCES1. Garrigue S, Bordier P, Barold SS, Clementy J. Sleep apnea: a new indication for cardiac pacing? Pacing Clin Electrophysiol 2004;27:204-11.
2. Choi SJ, Choi SB. Clinical feature of heart rate variability in obstructive sleep apnea patients. Korean J Sleep Med 2006;8:14-9.
3. Porges SW. Orienting in a defensive world: mammalian modifications of our evolutionary heritage. A Polyvagal Theory. Psychophysiology 1995;32:301-18.
4. Porges SW. The polyvagal theory: phylogenetic substrates of a social nervous system. Int J Psychophysiol 2001;42:123-46.
5. Yun AJ, Lee PY, Doux JD, Conley BR. A general theory of evolution based on energy efficiency: its implications for diseases. Med Hypotheses 2006;66:664-70.
6. Yun AJ, Lee PY, Doux JD. A paradigm for viewing biologic systems as scale-free networks based on energy efficiency: implications for present therapies and the future of evolution. Med Hypotheses 2006;67:651-7.
7. Aydin M, Altin R, Ozeren A, Kart L, Bilge M, Unalacak M. Cardiac autonomic activity in obstructive sleep apnea: time-dependent and spectral analysis of heart rate variability using 24-hour Holter electrocardiograms. Tex Heart Inst J 2004;31:132-6.
8. Yang A, Sch├żfer H, Manka R, Andri├® R, Schwab JO, Lewalter T, et al. Influence of obstructive sleep apnea on heart rate turbulence. Basic Res Cardiol 2005;100:439-45.
9. Szollosi I, Krum H, Kaye D, Naughton MT. Sleep apnea in heart failure increases heart rate variability and sympathetic dominance. Sleep 2007;30:1509-14.
10. Karcz M, Chojnowska L, Zareba W, Ruzyłło W. Prognostic significance of heart rate variability in dilated cardiomyopathy. Int J Cardiol 2003;87:75-81.
11. Jouven X, Empana JP, Schwartz PJ, Desnos M, Courbon D, Ducimeti├©re P. Heart-rate profile during exercise as a predictor of sudden death. N Engl J Med 2005;352:1951-8.
Fig.┬Ā1.Heart rate nondipper: male 73-year-old, obstructive sleep apnea syndrome, 4% Oxygen desatuation index 29, SDNN 78 ms, sleep heart rate 76 (arrow: Sleep heart rate). 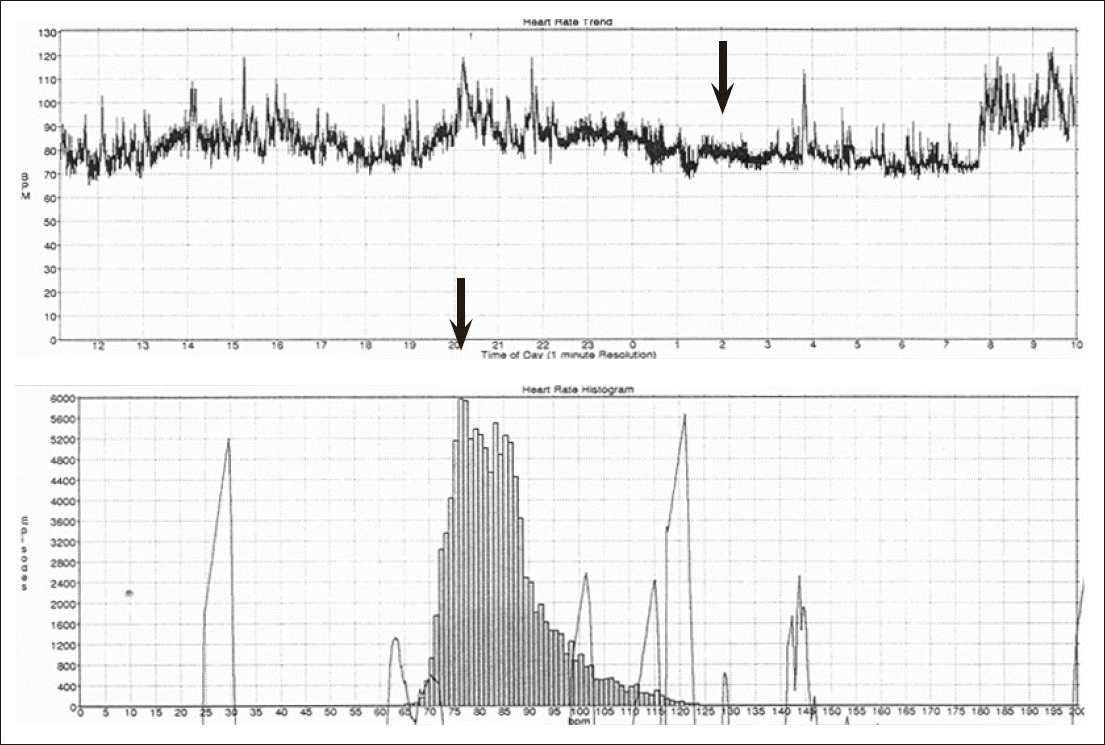
Fig.┬Ā2.Heart rate dipper: male 47-year-old, Obstructive sleep apnea syndrome, 4% Oxygen desatuation index 38.8, SDNN 111 ms, sleep heart rate 60 (arrow: Sleep heart rate). 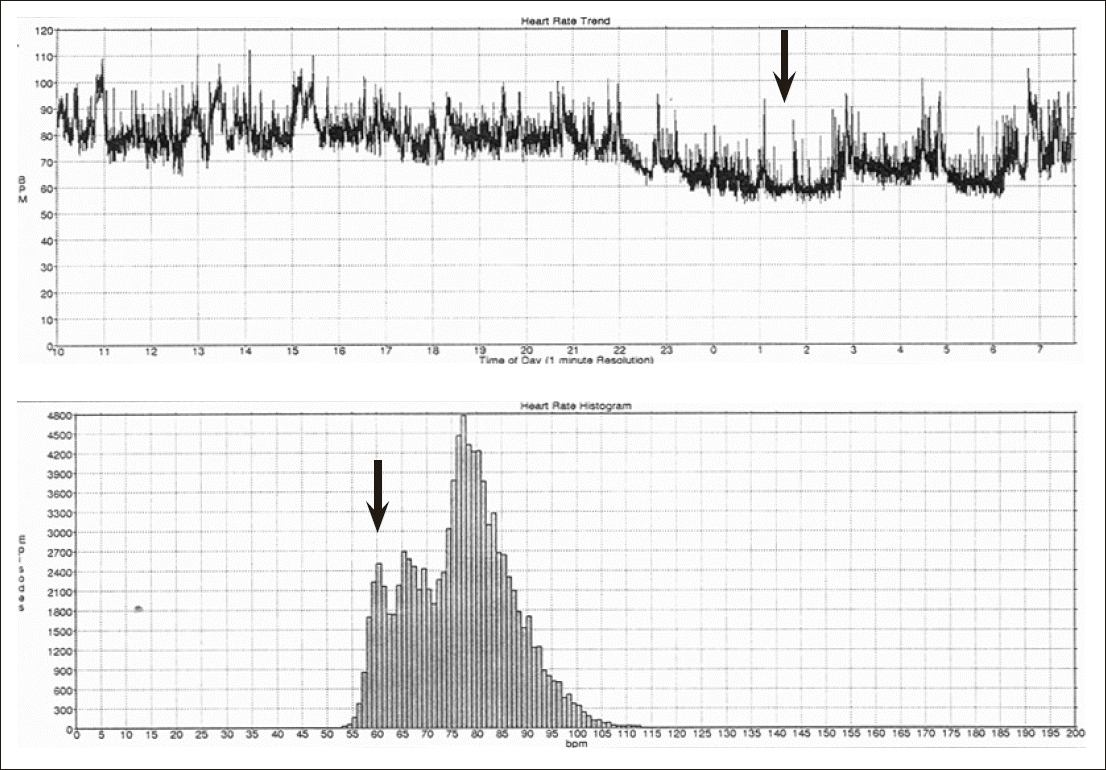
Fig.┬Ā3.Standard deviation of all normal R-R intervals (ms) in heart rate nondipper and dipper (mean + SD)(71.72 + 25.01 vs. 121.73 + 25.48, p < 0.0001). 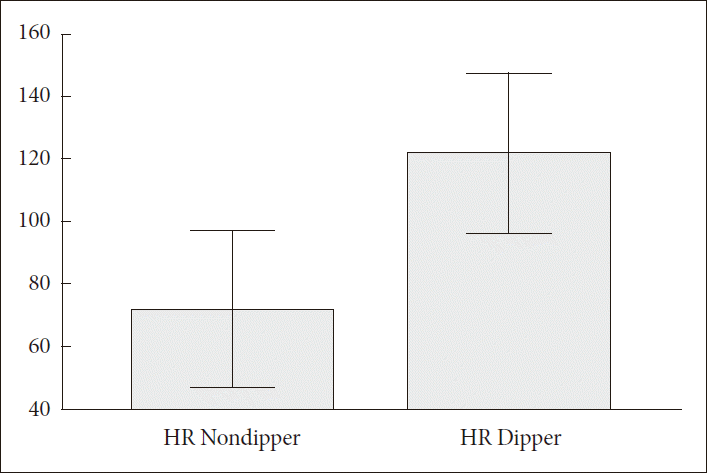
Fig.┬Ā4.Sleep heart rate (/min) in HR Nondipper and Dipper (mean + SD)(81.56 + 18.39 vs. 64.41 + 7.48, p = 0.0003). 
Fig.┬Ā5.Mean SaO2 in heart rate nondipper and dipper (mean + SD) (90.98 + 4.58 vs. 93.58 + 1.37, p = 0.0158). 
Table┬Ā1.HRV parameters in OSA and control subjects HRV: heart rate vatiability, OSA: obstructive sleep apnea, MeanNN: mean value of all normal-to-normal interbeat intervals, SDNN: standard deviation of all normal R-R intervals, SDANN: standard deviation of the average normal R-R intervals for each 5-min period, pNN50%: percentage of adjacent cycles that are > 50 ms apart, rMSSD: root mean square successive differences in milliseconds, LHR: ratio of low to high frequency, SHR: basal heart rate during sleep. Table┬Ā2.Differences between HR nondippers (n = 18) and dippers (n = 22) Table┬Ā3.HRV parameters in HR nondippers (n = 18) and dippers (n = 22) HRV: heart rate vatiability, MeanNN: mean value of all normal-to-normal interbeat intervals, SDNN: standard deviation of all normal R-R intervals, SDANN: standard deviation of the average normal R-R intervals for each 5-min period, pNN50%: percentage of adjacent cycles that are >50 ms apart, rMSSD: root mean square successive differences in milliseconds, LHR: ratio of low to high frequency. Table┬Ā4.Correlations between SDNN, SHR and ODI4 in HR nondipper and dipper subgroups SDNN: standard deviation of all normal R-R intervals, SHR: basal heart rate during sleep, ODI4: 4% oxygen desaturation index, HR: heart rate, ESS: Epworth sleepiness scale, BMI: body mass index, NC: neck circumference, PRR: pulse rate rise, MeanNN: mean value of all normal-to-normal interbeat intervals, SDANN: standard deviation of the average normal R-R intervals for each 5-min period, pNN50%: percentage of adjacent cycles that are >50 ms apart, rMSSD: root mean square successive differences in milliseconds, LHR: ratio of low to high frequency, SHR: basal heart rate during sleep. |
|
||||||||||||||||||||||||||||||||||||||