INTRODUCTION
Obstructive sleep apnoea (OSA) is a chronic disease that has exhibited increasing prevalence trends since the turn of the century [1,2]. By definition, it is a sleep-related breathing disorder that occurs during sleep, in which a partial or complete halt of airflow occurs despite ongoing respiratory effort [2-6]. Unfortunately, this disorder remains undiagnosed in a substantial proportion of the population [6-9], making it crucial for all healthcare professionals to increase awareness in society and to screen individuals exhibiting signs and symptoms related to OSA.
Due to the various factors that contribute to OSA, it is essential to characterise the upper airway (UA) in relation to the surrounding tissues, anatomical structures, neuromuscular function, and facial skeletal morphology as potential risk factors for OSA.
This study focused on the understanding of the functional mechanism(s) of the UA and facial skeletal anatomy as the anatomical phenotype of OSA in its pathogenesis.
STRUCTURAL AND PHYSIOLOGICAL DETERMINANTS OF OSA
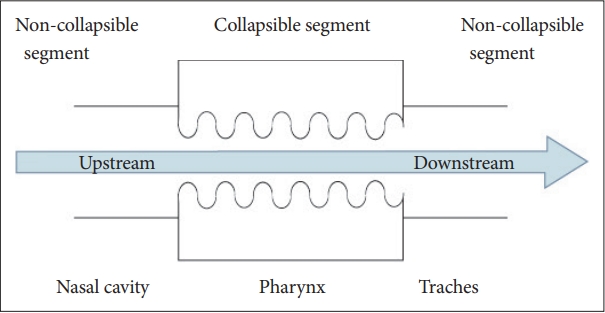
OSA is characterised by recurrent episodes of UA collapse during sleep [8,10,11]. It can occur at multiple levels at the site of the nose, nasopharynx, oropharynx, epiglottis, or hypopharynx in a different proportion in individual patients [12-15]. According to previous studies, the Starling resistor model describes the UA physiology (Fig. 1) [16-19].
Nasal Region
The nose is a non-collapsible structure and the initial entry site for air. It accounts for approximately 50% of the total airway resistance of the UA [17,18]. The nasal region, especially the internal nasal valve that comprised of septum, upper lateral cartilage, inferior turbinates, and nasal floor are the most important sites. Given that these entities comprise the narrowest portions of the nasal airway, aggravation of nasal obstruction is often found in OSA patients [20]. The nasal region in OSA can be explained by increased nasal airway resistance and the switch from nasal to mouth breathing due to nasal obstruction and impaired nasal ventilatory reflex.
Increased nasal airway resistance
The Starling resistor model reflects airway obstruction in accordance with an increase in nasal airway resistance. According to this model, suction forces are generated from the nose to the trachea during inhalation. An increase in nasal resistance generates greater negative pressure. However, the nose is a solid tube and will not collapse; as such, the collapsible pharyngeal segment is affected [18]. On the other hand, reduced in nasal resistance has been reported to improve UA patency [21,22].
Switch from nasal to mouth breathing
One of the various factors in the nasal region that contributes to the development of OSA is the switch from nasal breathing to oral breathing. This condition commonly occurs during nasal obstruction caused by narrowing of the internal nasal valve area [17,18]. For sleeping subjects, UA resistance is greater in those breathing through their mouth than their nose [23]. On the other hand, for subjects that are awake, the UA resistance is equal for both forms of breathing [23]. In addition, mouth breathing narrows the pharyngeal lumen, reduces retroglossal diameter due to further posterior displacement of the tongue, increases soft palate length, and oscillates redundant pharyngeal tissues [17,18, 24-26]. Furthermore, prolonged mouth breathing may lead to facial changes which can lead to a narrow nasal floor and deviated nasal septum [27].
Impaired or absent nasal ventilatory reflex
Impaired or absent nasal ventilatory reflex occurs in mouth breathers. Mouth breathing reduces nasal receptor activation and subsequently impairs the nasal ventilatory reflex [28]. Impaired nasal ventilatory reflex reduces spontaneous ventilation [23,28] causing failure of activation of the UA dilator muscles [23]. This condition worsens apnoea episodes because UA dilator muscle tone is inhibited, consequently reducing breathing and minute lung ventilation [16,17,24,29].
Nitric oxide
Nitric oxide (NO) is believed to play a role in the pathogenesis of OSA and snoring; however, its mechanism of action is not well understood [30]. NO is produced in a significant amount within the nose and paranasal sinuses, and plays a role in perpetuating UA patency [17]. It functions as a potent bronchial dilator and increases blood oxygen content by reducing perfusion-ventilation mismatch [17,31]. Furthermore, It plays a role in managing muscle tone, neuromuscular control of the pharyngeal musculature, sleep, and regulation of breathing [17,31]. With all the known functions of NO, the nasal region is a factor in the pathogenesis of OSA [32,33]. Therefore, this confirms that nasal obstruction is associated with snoring and OSA. In the presence of nasal obstruction, an increase in nasal resistance will occur, which may indirectly decrease the amount of NO and, therefore, nasal ventilation [32].
Soft Tissue Morphology in OSA
Fat deposition in the structure(s) surrounding the UA, such as the tongue, the soft tissue around the pharyngeal region, and pharyngeal muscles, can reduce the space occupied by the UA in those with OSA, especially in obese patients [34]. Changes in the anatomical phenotype of the soft tissue in OSA patients can effect physiological changes. Fig. 2 is a representative depiction of an unfavourable anatomy of the UA, which predisposes the patient to the development of OSA.
Soft palate and retropalatal region
Anatomically, the morphology of the soft palate in OSA patients appears to be thicker, longer, and increased in height, thus contributing to reduced airway dimensions [35-37]. The soft palate appears to be angled and thickened in this patient population [37,38]. In obese individuals, there is excessive deposition of the parapharyngeal fat pad extending to the submandibular space outside the maxillomandibular bony enclosure [39-42]. The presence of this excessive fat pad can further lead to concentric narrowing of the retropalatal pharynx, which consequently increases susceptibility to OSA [40,42]. Various researches have explained this phenomena as follows: increased mechanical load on the pharyngeal wall by the deposition of fat [43]; failure of the pharyngeal wall to sustain a higher passive critical closing pressure (Pcrit) than usual [44]; and an increase in collagen type 1 in the extracellular matrix of the lateral pharyngeal wall, which can lead to impairment or delay relaxation of the superior pharyngeal constrictor muscles [45].
Tongue
The anatomy of the tongue in this patient population is somewhat contributory to OSA, owing to an increase in tongue volume or mass [34,46,47] and its more posterior position [48,49]. These combinations lead to significant retropalatal and retroglossal narrowing [50]. A study by Wang et al. [49], reported that weight loss had an effect in reducing the volume of several UA soft tissues, mainly tongue fat, in obese and OSA patients.
The physiological effect of the tongue in OSA is mainly correlated with tongue fat and the genioglossus (GG) muscle. During sleep onset, GG muscle activity reduces in both OSA patients and healthy individuals [11,51,52]. However, given the anatomical changes in OSA, this reduction will further exacerbate UA collapse. In individuals without OSA, negative airway pressure, hypoxia, and recurrent hypercapnia increase GG activity. However, this is usually insufficient to open the UA for adequate ventilation due to poor muscle responsiveness [11,51,52]. Eventually, this leads to arousal from sleep and, thus, elevated loop gain to increase the ventilatory drive to reopen the airway [53,54]. This could be due to various factors including: inadequate muscle recruitment in the GG muscles, thus requiring a higher level of respiratory stimuli [6,55]; poor neural response to GG muscles [6,55]; and distorted orientation of the muscle fibres and number of fast fibres in the tongue in patients with OSA, which are easily fatigued [6,56]. All of these factors contribute to reduced stiffness in the GG and, subsequently, lead to an increase in compliance to UA collapse. These factors are independent of body mass index (BMI) in patients with OSA [57].
Facial Skeleton
The facial skeleton is essential to airway patency. According to previous studies, the soft tissue within the maxillomandibular region and the bony structure enclosing it affects the size of the pharyngeal airway [41,58,59].
Maxilla and mandible
Facial skeletal abnormalities, such as a small maxilla and/or mandible, are commonly encountered in patients with OSA. A small maxilla and mandible lead to a posteriorly positioned maxilla with a steep mandibular plane, leading to high upper and lower facial heights [41,50,60]. Due to these changes, there is overcrowding of the teeth by overeruption of the maxillary and mandibular teeth with proclined incisors. The position of the mandible determines the position of the tongue. Therefore, if the mandible and maxilla have a steep projection, both soft tissues (i.e., tongue and soft palate) are affected. All of these changes subsequently reduce available space for the UA [52].
A randomised case-controlled study by Suntherland et al. [61] recruited 363 OSA patients, 200 of whom were Chinese and 163 were Caucasian. According to the study, patients with OSA had reduced maxillary and mandibular depth angles. There were no interaction effects with sex or ethnicity, which means that these may be common OSA features in all groups. Furthermore, in a randomised case-controlled study by Tsuiki et al. [41], who compared the balance of craniofacial and tongue size between 50 OSA and 55 non-OSA subjects, the results revealed that patients with OSA had a significantly larger tongue, given the matched maxillomandibular dimension size. However, although the bony structure of the maxilla and mandible may limit soft tissue expansion, caudal expansion of the soft tissue in the oropharyngeal region remains possible [41].
Hyoid bone
The hyoid bone appears to be more inferiorly and posteriorly positioned in patients with OSA [38]. Since the hyoid bone is free and mobile and is not attached to other bones, expansion of the soft tissue caudally from the maxillomandibular bony enclosure may explain the shift of the hyoid bone caudally [58]. Furthermore, an increase in the distance between the hyoid and mandibular plane is of paramount consideration for OSA severity due to the attachment of the lingual musculature [52,62]. According to Starling model, increasing the distance increases the airway length, leading to instability [52,63]. In addition, the musculature attachments pull the tongue backward, causing narrowing of the pharyngeal airway [38,41,58,64].
RECENT RESEARCH TRENDS INVESTIGATING UA ANATOMY IN OSA
This study focused on three recent research trends in OSA: pathophysiological phenotypes of OSA and anatomical traits; phenotype labelling with drug-induced sleep endoscopy (DISE) in patients with OSA; and non-invasive diagnostic tools for identifying anatomical factors affecting OSA.
Pathophysiological Phenotypes of OSA and Anatomical Traits
Understanding OSA phenotype is an important tool for designing strategies to identify and target interventions for patients at risk. Four important pathophysiological phenotypes of OSA have been discussed in most literature reviews [11,51,55,65,66]: UA collapsibility, UA anatomy response to changes in Pcrit; muscle responsiveness, the capability of the UA dilator muscles to react to pharyngeal collapse (muscle responsiveness); low arousal threshold, wakefulness before muscle activation; and high loop gain and ventilatory loop gain changes in response to blood-gas disturbances during sleep. Given that the pathogenesis of OSA is multifactorial, it is important to understand the potential development of OSA using a combination of pathophysiological phenotypes and an anatomical predisposition [6,51,55,67].
Generally, the UA in patients with OSA is usually smaller or narrowed compared to that in healthy individuals [40,63,68]. Furthermore, the soft tissues of the UA are unorganized. These factors lead to an increase in Pcrit and a predisposition to pharyngeal collapse [69]. Pierce et al. [26] reported that UA dilator muscle activation is directly proportional to Pcrit changes in awake OSA patients. The GG muscles activity was weakened with sleep onset in patients with OSA. Owing to the weakened response of the UA dilator muscles, UA patency is prone to collapse [11,51,52]. Subsequently, this would lead to arousal from sleep in patients with mild-to-moderate OSA with a low arousal threshold in response to increased negative pressure, hypoxia, or hypercapnia [56,70-72]. The feedback would stimulate high ventilatory loop gain to stabilize airway status [53,54]. A condition predominantly associated with severe OSA, in which patients exhibit a high arousal threshold and fail to respond to these stimuli [70], is believed to be due to neural plasticity in response to repetitive episodes of hypoxaemia [53]. They are more prone to hypoxia due to prolonged apnoea. In this condition, the high loop gain would be exaggerated due to the response of prolonged apnoea leading to hyperventilation to combat blood-gas instability [53,54,66].
Phenotype Labelling Using DISE in OSA
The repetitive collapse of airway structures causes OSA. The site of obstruction can be identified using DISE, and each obstruction site may be correlated with an increase in OSA severity. However, some recent studies have reported a negative correlation between a specific type of anatomical obstruction and OSA severity [15,73,74]. This leads to the question of whether the collapse site identified using DISE can be regarded as a phenotype predicting overall disease severity.
Velum
Concentric narrowing in the retropalatal region (Vccc) is associated with the parapharyngeal fat pad [40]. Jang et al. [40] reported that the parapharyngeal fat pad does not act as a lateral mass, but as an increase in pharyngeal wall redundancy, which subsequently leads to an increase in surrounding pressure in the retropalatal region.
Hypoglossal nerve stimulation therapy is contraindicated in patients with Vccc identified on DISE [68,75]. However, Vccc exhibited poor response to surgical treatment [75-77]. Vccc and complete lateral collapse at the oropharyngeal level predict poor outcome(s) and treatment response in patients with mild-to-moderate OSA undergoing mandibular advancement device (MAD) therapy [78]. On the other hand, those with Vccc benefited from maxillomandibular advancement surgery in a prospective study [79]. Patients with Vccc required lower continuous positive airway pressure (CPAP) than those with anteroposterior velum collapse [80].
Oropharynx
Oropharyngeal lateral collapse is caused by thickening of the lateral pharyngeal wall and increase in the volume of the parapharyngeal fat pad [81,82]. A retrospective clinical review by Soares et al. [82] revealed that approximately 53% of OSA patients exhibited significant lateral oropharyngeal collapse at the retrolingual level, and that it was primarily observed in males, those with a higher BMI, and higher apnoea-hypopnoea index (AHI) than those who did not exhibit collapse. These findings corresponded with those reported in a cohort study by Schellenberg et al [83]. Furthermore, the study reported that enlargement or hypertrophy of the oropharyngeal soft tissue structures, such as the tonsils, soft palate, uvula, and tongue, also mediate the narrowing of the oropharyngeal airway [83].
There is a male predominance in oropharyngeal collapse; which according to a previous study, is hormonal [84]. Females exhibit decreased soft tissue volume in the neck and parapharyngeal fat despite higher percentages of body fat levels [83,85]. Females also exhibit increased pharyngeal dilator muscle activity. Thus, they exhibit a lower propensity for UA collapsibility than males; however, there are limited data supporting this assertion.
Tongue
The tongue is the most important anatomical factor in OSA [48]. The effect of obesity has been reported to be dependent on tongue volume [49]. Thus, tongue base collapse and BMI are believed to be positively correlated. However, a recent DISE study revealed that tongue base obstruction and BMI were negatively correlated [88]. The study reported that tongue base obstruction was observed in all patients with low BMI (100%) in the supine position [73]. This study highlighted an important clinical characteristic of OSA patients with a low BMI. In addition, these patients exhibited unfavourable characteristics for CPAP use [80] but demonstrated a good response to head rotation or MAD therapy [15,78].
Epiglottis
The epiglottis is typically positioned in an upright position to facilitate air flow. Epiglottic “trapdoor” or floppiness has been reported in adults with sleep-related breathing disorders [14,89,90], and can be identified using DISE. Nevertheless, Azarbarzin et al. [91] recently reported that discontinuity and “jaggedness” of airflow are the trademarks of epiglottic collapse in patients with OSA. Epiglottis collapse is exacerbated by the application of CPAP [74,89,92]. In such a situation, partial epiglottectomy has been shown to improve residual AHI [74]. In addition, patients with epiglottic collapse exhibited less severe OSA and lower BMI [15, 88]. Thus, they demonstrated a good response to non-CPAP treatments such as positional therapy or MAD [14,15,88]. However, the pathophysiology regarding how and why it occurs remains controversial.
RECENT UPDATE REGARDING NON-INVASIVE DIAGNOSTIC TOOLS FOR IDENTIFYING ANATOMICAL FACTORS IN OSA
The development of non-invasive tools to diagnose OSA is a topic of ongoing research and has resulted in new and emerging technologies [46,93,94]. Huang et al. [46] examined the impact of dynamic changes in tongue base thickness (TBT) in OSA during awake and sleep using DISE ultrasonography. Results revealed that TBT during DISE ultrasonography was significantly higher in sedated sleep state than that during the awake state and was positively correlated with AHI. This was the first study to demonstrate the feasibility of applying DISE ultrasonography to detect UA changes in patients with OSA.
Furthermore, Op de Beeck et al. [95] used a pneumotachometer to investigate flow measurement and performed shape analysis during DISE to identify the sites of UA collapse. The study revealed that patients with epiglottic collapse had significantly higher negative effort dependence (NED) values than those without epiglottic collapse. The breaths in epiglottic collapse have significantly higher NED than those in complete oropharyngeal, tongue base, or lateral pharyngeal wall collapse.
Various studies have used commercially available software to perform computational fluid dynamics (CFD) analysis of the human UA. CFD can integrate structural analysis software using a fluid-structure interaction simulation to tackle human UA problems which involve movement, motion, and deformation of soft tissue or airway wall. This approach can be used to analyse and detect airway collapse during breathing [93]. In conjunction with CFD, MRI has been used to determine relationships between the retropalatal airway, pharyngeal length, and craniofacial structures in patients with OSA [94].