Many chronic insomnia patients use sleep medication well beyond the recommended duration, and patients who are hypnotic-dependent encounter several challenges when attempting to discontinue. The current article presents these challenges and reviews studies that have added cognitive-behavioral therapy for insomnia (CBTI) to a gradual tapering program to facilitate discontinuation of hypnotic medication in chronic insomnia patients. The current literature provides substantial support for using CBTI to facilitate discontinuation of hypnotic medication in chronic insomnia patients. Across several randomized clinical trials, the majority of the findings indicate that using CBTI, delivered by a therapist or self-help, along with a supervised or gradual tapering program is superior to tapering only or CBTI only in terms of medication discontinuation. Although these results are promising, it should be noted that the long-term evidence is more equivocal, as over 40% of patients relapse in the need for sleep medication within 2 years, which may highlight the importance of adding follow-up booster sessions in hypnotic-dependent insomniacs. These findings indicate that rather than choosing between CBT and hypnotic medication, clinicians might be better served using both treatment modalities to optimize the relative strengths of each treatment. Such an approach represents a more modern view of treating chronic insomnia patients by using both pharmacological and non-pharmacological tools.
INTRODUCTION
Insomnia is a highly prevalent disorder that affects 10–15% of the general population.1,2 Chronic insomnia is associated with significant morbidity, such as reduced quality of life, increased risk for psychiatric disorder, and higher risk for various physical diseases such as hypertension and diabetes.1,3–7 Over the past 20 to 30 years, several clinical trials have examined the efficacy of various treatments for insomnia. The accumulated research evidence supports the efficacy of both pharmacological and non-pharmacological treatments for insomnia, yet each of these treatment modalities has limitations in clinical practice. A sophisticated approach that has gained research attention and appears to be translatable to real-world clinical practice is to combine a pharmacological treatment with a non-pharmacological treatment. Specifically, cognitive-behavior therapy for insomnia (CBTI) has been used to aid in the discontinuation of chronic hypnotic use among those who began using hypnotics and have become chronic users. The aims of this paper are to present the issues related to chronic hypnotic use and to review the literature on the addition of CBTI to facilitate the discontinuation of hypnotic medication in chronic insomnia patients.
Pharmacological Treatment of Insomnia: Hypnotic Medication
Historically, pharmacotherapy has been the first line treatment for insomnia and remains a popular choice of treatment.8,9 Approximately 3 to 10% of the general population use hypnotic medication, and this rate increases to 20% among older individuals.10–13 Overall, studies have demonstrated that hypnotics can improve sleep by reducing sleep onset latency and increasing total sleep time and these effects occur within the first week of initiating treatment.14–16 Currently, the most commonly used medications for insomnia include benzodiazepine receptor agonists (BzRAs), melatonin receptor agonists, and antidepressants.17 There are now 10 medications approved by the Food and Drug Administration to treat insomnia (
A Pattern of Hypnotic Dependency
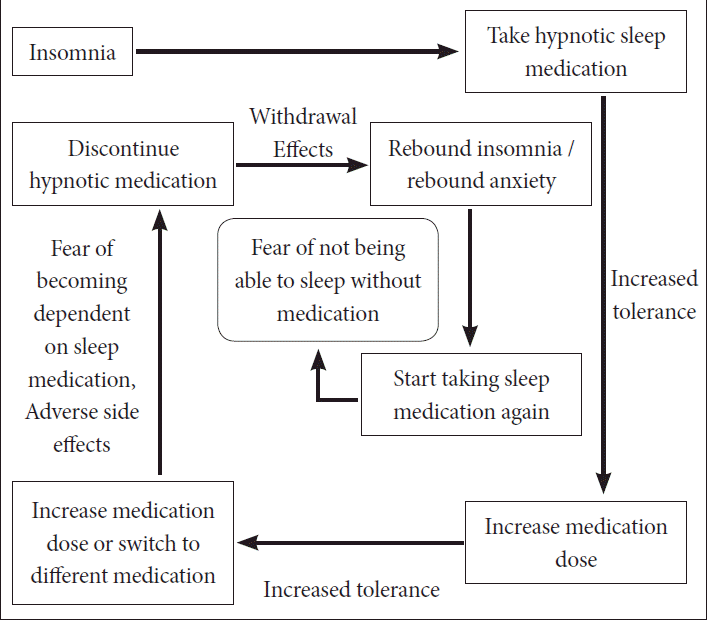
Chronic insomnia patients who are hypnotic-dependent encounter several challenges when attempting to discontinue (
Non-Pharmacological Treatment of Insomnia: Cognitive-Behavioral Therapy for Insomnia (CBTI)
CBTI is a short-term treatment that is based on cognitive and behavioral principles to treat insomnia. The main purpose of CBTI is to eliminate maintaining factors that are presumed to perpetuate chronic insomnia. CBTI usually consists of 4–8 sessions and utilizes a multicomponent approach that includes sleep hygiene, sleep restriction, stimulus control, relaxation training, and cognitive therapy.28
Review of Studies Combining CBTI with Sleep Medication Discontinuation
In addition to CBTI as a primary treatment, the AASM practice parameters also recommend the use of CBTI in treating insomnia patients who were chronic hypnotic users.30 Given the potential risk of long-term hypnotic use, clinicians are often met with the challenge of how to help their patients discontinue hypnotic medication while managing their insomnia. One novel approach has been to combine a non-pharmacological treatment, such as CBTI, to aid in the discontinuation of sleep medication in chronic insomnia patients. There have been several uncontrolled studies or small clinical trials (n < 30), and two large scale randomized clinical trials that have investigated utilizing CBTI to facilitate discontinuation of hypnotic medication.
One of the first studies in this area was conducted by Morin et al.,35 who used CBTI combined with a supervised medication tapering schedule in a multiple baseline design with 5 insomnia patients who were dependent on benzodiazepines (alprazolam, diazepam, lorazepam) at least 4 nights per week (range of duration for benzodiazepine use = 2–15 years). Among the 5 participants, 4 were successful in discontinuing medication within 6–8 weeks, and 3 patients remained drug-free at 3-month follow-up. While sleep efficiency decreased from baseline to the end of the medication withdrawal phase, sleep efficiency improved at 3-month follow-up. This seminal study demonstrated the potential effects of combining CBTI with a medication tapering program, but the sample size and methodological rigor was very limited.
Riedel et al.36 examined 21 medicated and 20 non-medicated individuals with insomnia who were participating in a sleep medication withdrawal program that provided psychoeducation about a gradual tapering system. The medicated insomniacs were using sleep medication at least 6 times per week for at least 3 weeks (mean duration 4.7 years). Sleep medications included benzodiazepines (alprazolam, lorazepam, temazepam, triazolam, chloraepate, clonazepam, estazolam, quazepam) and/or antidepressants (amitriptyline, doxepin, trazodone, imipramine) as well as over-the-counter medication. Half of the participants in the sleep medication group and half of the participants in the nonmedication group received stimulus control treatment in addition to the withdrawal program. Medication consumption was defined in two ways - frequency of medication taken during the phase of the study, and percent reduction of medication dosage from baseline to posttreatment, and posttreatment to follow-up. Results from their study indicated that participants were using sleep medication an average of 6.2 nights per week at baseline, which reduced significantly to 1.7 nights per week through a gradual withdrawal program, and participants reduced their medication dosage by 76.6% without experiencing aversive sleep deterioration. However, at follow-up, some relapse was reported as medicated participants increased their frequency of sleep medication use to 3.1 nights per week, with increases in dosages. Although stimulus control treatment did not improve patient’s ability to reduce sleep medication, adding a stimulus control treatment to a gradual tapering program improved sleep and daytime functioning after medication reduction, compared to those who did not receive stimulus control treatment. Additionally, this study also showed the difficulty of sustaining treatment effects long-term, which indicates the importance of having follow-up booster sessions to continue treatment effects.
In testing another component of CBTI, Lichstein et al.37 explored the effectiveness of adding relaxation to assist in sleep medication withdrawal. Forty participants (only half using chronic sleep medication) participated in the study, and half of the medicated participants and unmedicated participants received progressive muscle relaxation. The medicated participants were taking benzodiazepines, antidepressants, and over-the-counter medication (mean duration = 5.9 years). All medicated participants received a gradual sleep medication tapering program, and sleep medication consumption reduced by nearly 80% at post-treatment. Participants who received relaxation therapy in addition to the tapering program obtained additional benefits in sleep efficiency, sleep quality, and reduced withdrawal symptoms. Relaxation therapy benefited both medicated and nonmedicated participants, and the authors concluded that adding relaxation therapy in addition to a medication tapering program may be beneficial for hypnotic-dependent insomniacs.
Using improved methodology, two randomized clinical trials have investigated the effectiveness of CBTI in the context of hypnotic discontinuation. First, a study by Morin et al.38 conducted a randomized clinical trial comparing a supervised tapering program, CBTI, or combined treatment (supervised tapering with CBTI) on 76 individuals with long-term benzodiazepine use (mean duration 19.3 years). While all three interventions significantly helped reduce both the quantity and frequency of benzodiazepine use after the 10-week intervention period, individuals who received a combination of both supervised tapering and CBTI had significantly higher proportion of individuals who remained benzodiazepine-free at post-treatment (85%) compared to those who received a medication taper alone (48%) or CBTI alone (54%). Individuals who received both CBTI and medication tapering, or CBTI alone had greater subjective sleep improvements compared to individuals who received a medication taper alone. This study provided more definitive evidence that the combination of CBTI with a structured tapering program is needed to obtain the optimal outcome for hypnotic discontinuation.
In addition to the acute effects, In addition to the acute effects, Morin et al.41 also examined the long-term effects and relapse rates of individuals from the study above38. While there was a high rate of relapse for sleep medication across all three conditions (42.6%), participants who either received a supervised medication taper (30.8%) or combined approach (33.3%) had significantly less relapse rates compared to the CBTI only group (69.2%). Additionally, time to relapse was significantly longer for both the medication taper only group (18.6 months) and combined treatment (12.6 months) compared to the CBTI only group (8.5 months). Similar to the earlier findings of Riedel and colleagues (1998), the short-term benefits of these interventions are somewhat diminished by the high relapse rates and suggest that CBTI booster sessions may be useful in preventing relapse of hypnotic use.
Another randomized clinical trial by Belleville et al.10 extended the Morin et al.38 study by testing a self-help CBTI program to see if it a more conservative method of CBTI that uses fewer resources could also produce similar results. In this study, an 8-week hypnotic taper program alone was compared with the tapering program combined with a self-help CBTI program in 53 chronic users of hypnotics (benzodiazepines, zopliclone, zaleplon) whose average use of sleep medication was 6.75 nights per week (mean duration = 14.1 years). Both groups decreased frequency (less than once a week) of hypnotic use, and nightly dosage (lorazepam equivalent) decreased from 1.67 mg to 0.12 mg. Individuals who received CBTI combined with the tapering program showed improvements in sleep efficiency and total wake time, compared to those who received a tapering program alone. These findings suggest that the addition of a self-help CBTI to a tapering program over 8 weeks could be an alternative and cost-effective option for individuals who might not have access to a CBTI therapist.
In addition to the promising outcome evidence indicating that the addition of CBTI to a gradual tapering program leads to improvements in insomnia symptoms as well as reductions in the frequency and dosage of medication, another group of studies have examined baseline symptomotology and psychological factors that can predict successful medication discontinuation. Belanger et al.39 conducted secondary analyses from the Morin et al.38 dataset and found that compliance with treatment and success in reducing hypnotic medication was significantly associated with self-efficacy. Belleville and Morin40 also conducted secondary analyses of the Belleville et al.10 study comparing a hypnotic taper program with or without self-help CBTI. They reported that while there were no significant psychological differences at baseline between medication-free individuals and those still using medication at the end of a taper intervention, group differences emerged midway during treatment. Participants who remained medication-free at 6-month follow-up had less severe insomnia and anxiety symptoms, more positive perceived health and higher self-efficacy about refraining from using hypnotics at 6-month follow-up. Together, these findings suggest that severity of insomnia, self-efficacy, and perception of health are important factors to evaluate in determining candidates who may need more intensive and individualized therapeutic attention to target these factors, which would subsequently lead to higher likelihood in benefitting from using CBTI as part of a medication tapering program.
Finally, a recent study by Morin et al.41 examined different sequential pathways of CBTI only, and CBTI plus zolpidem. The aim of this study was not specifically on medication tapering, but rather to test different sequences of initiating and tapering pharmacological and non-pharmacological treatments. In terms of short-term outcomes at 6 weeks, the rates of remission and response for CBTI only was found to be similar to CBTI plus zolpidem. However, in terms of long-term outcomes at 6 months, the combined treatment was superior to CBTI alone, with the best outcome achieved by using CBT plus zolpidem for the first 6 weeks, followed by CBTI alone during the maintenance phase. A key strength of this study is that the treatment sequences are similar to real-world decisions made in the clinic and thus can be more readily implemented into clinical practice.
CONCLUSION
Based on the literature reviewed, there appears to be substantial support for using CBTI to facilitate discontinuation of hypnotic medication in chronic insomnia patients. Across several randomized clinical trials, the majority of the findings indicate that using CBTI, delivered by a therapist or self-help, along with a supervised or gradual tapering program is superior to tapering only or CBTI only in terms of medication discontinuation and insomnia symptoms. The findings also indicate that patients who have more severe insomnia, a more negative perception of health, and lower self-efficacy after treatment are more likely to have difficulty in discontinuing hypnotics compared to patients who do not exhibit these characteristics after receiving treatment. This indicates that these individuals may need more intensive and individualized therapeutic attention following treatment to benefit from receiving CBTI in addition to a medication tapering program. Although these results are promising, it should be noted that the long-term evidence is more equivocal, as over 40% of patients relapse in the need for sleep medication within 2 years, which may highlight the importance of adding follow-up booster sessions in hypnotic-dependent insomniacs.
These findings have important implications for optimizing patient care in the sleep clinic. Rather than choosing between CBT and hypnotic medication, clinicians might be better served using both treatment modalities to optimize the relative strengths of each treatment. Since the benefits of hypnotic medication occurs rapidly, within the first week, and CBTI has demonstrated long-term effects, a combination of the two approaches might yield both immediate and long-term relief from insomnia. Such an approach represents a more modern view of treating chronic insomnia patients by systematically using both pharmacological and non-pharmacological tools. It is hoped that further research and training of sleep clinicians will encourage the use of multidisciplinary teams that can integrate these treatments rather than choosing between the treatments in a limited clinical practice.
The authors have no financial conflicts of interest.
REFERENCES
Figure and Tables
Difficulty of discontinuing hypnotic medication in insomnia patients. From the educational material of “Introduction of Cognitive-Behavioral Therapy for insomnia” by S. Chung, in “Insomnia symposium 2010” held by The Korean Society of Sleep Medicine.
Selected drugs used for insomnia in the United States
| FDA-approved hypnotics
|
Drugs commonly used off-label
|
OTC medications
|
||||
|---|---|---|---|---|---|---|
| Benzodiazepines | Other benzodiazepine receptor agonists | Melatonin receptor agonist | Benzodiazepines | Antidepressants | Antipsychotics | Miscellaneous |
| Triazolam | Zaleplon | Ramelteon | Lorazepam | Trazodone | Olanzepine | Valerian |
| Temazepam | Zolpidem | Alprazolam | Mirtazapine | Quitiepine | Melatonin | |
| Estazolam | Zolpidem CR | Clonazepam | Doxepin | Alcohol | ||
| Flurazepam | Eszoplicone | Amitriptyline | Diphenhydramine | |||
| Quezapam | Cyclobenzaprine | |||||
| Hydroxyzine | ||||||
These constitute the most commonly used agents for the management of insomnia in the United States, either by prescription or over the counter (OTC). Among those drugs with a Food and Drug Administration (FDA) indication for insomnia, flurazepam, and quazepam are not often used because of their long duration of action and predisposition to residual sedation. Among the drugs without an FDA indication for management of insomnia, doxepin and amitryptaline are less commonly used for their sedating effect, in comparison to the other sedating antidepressants listed.
Used with permission from “Efficacy of Hypnotic Medications and Other Medications Used for Insomnia” by Feren S, Katyal A, and Walsh JK. From Insomnia, Sleep Medicine Clinics, Volume 1 (3), 2006. Ed. Tom Roth.
Summary of cognitive-behavioral therapy for insomnia components
| Component | Treatment goal |
|---|---|
| Sleep hygiene | Eliminate habits that are counterproductive for sleep through education on general guidelines about health behaviors and environmental factors that negatively impact sleep (e.g., reduce drinking alcohol, avoid rigorous exercise close to bed time). |
| Stimulus control | Strengthening sleep cues (e.g., bed and bedroom) with sensations of sleepiness by limiting the amount of time spent awake in bed. Patients are instructed to only go to bed when sleepy, use the bed only for sleep and sexual activity, and leave the bed if unable to fall asleep for more than 15 to 20 minutes. |
| Sleep restriction | Restrict time in bed to improve sleep drive and consolidate sleep. By systematically reducing time in bed, homeostatic pressure for sleep is increased, , which in turn increases sleep consolidation. The goal of sleep restriction is to reach sleep efficiency of higher than 85% and to avoid compensating for poor sleep. |
| Relaxation techniques | Reduce physiological arousal and decrease anxiety (e.g., engaging in deep breathing exercises, progressive muscle relaxation, or autogenic training to reduce somatic tension or intrusive thoughts that interfere with sleep). |
| Cognitive therapy | Address and correct maladaptive thoughts, beliefs, and expectations about sleep that interfere with sleep and daytime functioning (e.g., misattributions of daytime impairments to poor sleep). |